Olmesartan medoxomil, tablets, 10 mg, 20 mg, and 40 mg, Olmetec®, November 2005
Public Summary Document for olmesartan medoxomil, tablets, 10 mg, 20 mg, and 40 mg, Olmetec®, November 2005
Page last updated: 27 February 2006
PDF version of this page (PDF 125 KB)
Public Summary Document
Product: Olmesartan medoxomil, tablets, 10 mg, 20 mg, and 40 mg, Olmetec®
Sponsor: Pfizer Australia Pty Ltd
Date of PBAC Consideration: November 2005
1. Purpose of Application
Request for listing of olmesartan medoxomil as an unrestricted benefit for the treatment
of hypertension.
2. Background
This drug has not previously been considered by the PBAC.
3. Registration Status
Olmesartan medoxomil was registered by the TGA on 6 September 2005 for the treatment
of hypertension.
4. Listing Requested and PBAC’s View
Unrestricted benefit.
The PBAC noted that an unrestricted listing was consistent with other PBS-listed angiotensin
II antagonists.
5. Clinical Place for the Proposed Therapy
This product will provide another angiotensin II antagonist to the currently listed
drugs, irbesartan, candesartan, eprosartan and telmisartan for the treatment of hypertension.
6. Comparator
The PBAC accepted the submission’s nomination of irbesartan as the appropriate comparator.
7. Clinical Trials
The submission provided (a) one head-to-head randomised trial comparing the fixed
starting doses of olmesartan 20 mg with irbesartan 150 mg in patients with essential
hypertension over 8 weeks and (b) an indirect comparison of eight randomised olmesartan
trials with ten randomised irbesartan trials, and involving placebo as the common
reference.
A list of published trials included in the submission is shown below.
| Trial/first author | Protocol title | Publication citation |
|---|---|---|
| 866-411/ Oparil et al |
Comparative efficacy of olmesartan, losartan, valsartan and irbesartan in the control
of essential hypertension. Use of 24-hour ambulatory blood pressure monitoring to assess antihypertensive efficacy: a comparison of olmesartan medoxomil, losartan potassium, valsartan and irbesartan. |
The Journal of Clinical Hypertension 2001; III:283-318. American Journal of Cardiovascular Drugs 2005; 5:41-50. |
| 866-204/ Neutel et al |
Antihypertensive efficacy of olmesartan medoxomil, a new angiotensin II receptor antagonist, as assessed by ambulatory blood pressure measurements. | Journal of Clinical Hypertension 2002; 4:325-31. |
| 866-415/ Chrysant et al |
Antihypertensive efficacy and safety of olmesartan medoxomil compared with amlodipine for mild-to-moderate hypertension. | Journal of Human Hypertension 2003; 17:425-32. |
| 866-318/ Chrysant et al |
Evaluation of antihypertensive therapy with the combination of olmesartan medoxomil and hydrochlorothiazide. | American Journal of Hypertension 2004; 17:252-9. |
| 037/ Kochar et al |
Matrix study of irbesartan with hydrochlorothiazide in mild-to-moderate hypertension. | American Journal of Hypertension 1999; 12:797-805. |
| Kassler-Taub et al | Comparative efficacy of two angiotensin II receptor antagonists, irbesartan and losartan, in mild-to-moderate hypertension. | American Journal of Hypertension 1998; 11:445-53. |
| Gradman et al | A novel orally effective renin inhibitor, provides dose-dependent antihypertensive efficacy and placebo-like tolerability in hypertensive patients. | Circulation 2005; 111:1012-8. |
| 038/ | Double-blind, placebo-controlled comparison of the combination of irbesartan and hydrochlorothiazide versus individual components in mild-to-moderate hypertension. FDA report (new drug application package) 1997. | Centre for drug evaluation and research application number; NDA 20757 and 20758. Medical review; 020757ap_avapro_medrP3: 222-6 [internet]. |
| 029/ Guthrie et al |
Efficacy and tolerability of irbesartan, an angiotensin II receptor antagonist in primary hypertension. A double-blind, placebo-controlled, dose-titration study. | Clinical Drug Investigation 1998; 15:217-27. |
| 030/ Fogari et al |
24-hour blood pressure control by once-daily administration of irbesartan assessed by ambulatory blood pressure monitoring. | Journal of Hypertension 1997; 15:1511-8. |
| 002/ | A multicenter, 8 week study of the antihypertensive activity, tolerability and safety of irbesartan in subjects with mild-to-moderate hypertension. FDA report (new drug application package) August 1997. Centre for drug evaluation and research application number; NDA 20757 and 20758. | Medical review; 020757ap_avapro_medrP2: 138-142; 222-6 [internet]. |
| 025/ Pool et al |
Dose-related antihypertensive effects of irbesartan in patients with mild-to-moderate hypertension. | American Journal of Hypertension 1998; 11:462-70. |
| 050 | A multicenter, 8 week study of the antihypertensive activity, tolerability and safety of irbesartan in subjects with mild-to-moderate hypertension. FDA report (new drug application package) August 1997. Centre for drug evaluation and research application number; NDA 20757 and 20758. | Medical review; 020757ap_avapro_medrP4: 246-7, 138-142, 222-6 [internet]. |
| Fogari et al | Effects of four angiotensin II-receptor antagonists on fibrinolysis in postmenopausal women with hypertension. | Current Therapeutic Research 2001; 62:68-78. |
8. Results of Trials
The submission relied on measures of change in blood pressure as the primary outcome
for analysis. The results of the key trial are summarised below.
|
Olmesartan 20mg (N=145) |
Irbesartan 150mg (N=145) |
p-value |
95% upper confidence limit |
|
|---|---|---|---|---|
| Change from baseline DBP mmHg mean (SD) LS mean |
-11.2 (8.8) -11.5 |
-9.7 (8.3) -9.9 |
0.0412 | -0.09 |
| Change from baseline SBP mmHg mean (SD) LS mean |
-11.4 (13.9) -11.3 |
-11.0 (13.6) -11.0 |
0.4254 | 2.24 |
DBP = diastolic blood pressure; SBP = systolic blood pressure; LS = least squares;
SD = standard deviation
The PBAC noted there was a statistically significant difference in change in mean
trough diastolic blood pressure favouring olmesartan, but this was not considered
to be a clinically important difference. There was no statistically significant difference
between olmesartan and irbesartan in the secondary analysis, change in mean trough
systolic blood pressure.
The PBAC noted that there appeared to be little difference in response between olmesartan
20 mg and 40 mg on the primary outcome, diastolic blood pressure in the indirect comparison.
The dose response curves with 95% CI for olmesartan and irbesartan calculated from
placebo-adjusted reductions in diastolic blood pressure using fixed effects meta-analytic
models are shown below.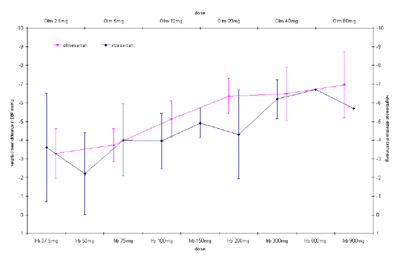
Dose response curves click to view full version. (GIF 11 KB)
The only adverse event that occurred in >1% of patients and at higher incidence than
placebo was dizziness (2.5% versus 0.9% respectively) for olmesartan and musculoskeletal
trauma (1.9% versus 0.5% respectively) for irbesartan in placebo-controlled trials
reported in the product information. Olmesartan and irbesartan appeared to have similar
toxicity profiles with incidence rates of adverse events comparable to placebo.
9. Clinical Claim
In the submission olmesartan was described as being no worse than irbesartan in terms
of effectiveness and toxicity. This was considered by the PBAC to be a reasonable
description.
10. Economic Analysis
Preliminary and modelled economic evaluations were not presented in the submission.
The choice of the cost-minimisation approach was considered valid by the PBAC.
The equi-effective doses in the context of cost-minimisation were olmesartan 10 mg
and irbesartan 75 mg, olmesartan 20 mg and irbesartan 150 mg and olmesartan 40 mg
and irbesartan 300 mg. This relativity was based on the head-to-head comparison of
olmesartan 20 mg versus irbesartan 150 mg and the dose response curves constructed
from meta-analyses of olmesartan versus placebo and irbesartan versus placebo. The
equi-effective doses proposed in the submission were considered by the PBAC to be
reasonable based on the level of evidence provided.
11. Estimated PBS Usage and Financial Implications
The submission estimated that the likely number of packs dispensed/year to be up to
between 700,000 and 800,000 in Year 4 of listing. The PBAC accepted that this was
a reasonable estimate.
The PBAC accepted the submission claim that olmesartan would not result in costs to
the PBS.
12. Recommendation and Reasons
The PBAC recommended listing on a cost-minimisation basis compared to irbesartan with
1 mg olmesartan being equivalent to 7.5 mg irbesartan.
Recommendation:
OLMESARTAN MEDOXOMIL, tablets, 10 mg, 20 mg and 40 mg
| Restriction: | Unrestricted benefit |
| Maximum quantity Repeats: |
30 (all strengths) 5 (all strengths) |
13. Context for Decision
The PBAC helps decide whether and, if so, how medicines should be subsidised in Australia.
It considers submissions in this context. A PBAC decision not to recommend listing
or not to recommend changing a listing does not represent a final PBAC view about
the merits of the medicine. A company can resubmit to the PBAC or seek independent
review of the PBAC decision.
14. Sponsor’s Comment
Notwithstanding the recommendation to list, the recent decline in the price of angiotensin II antagonists due to reference pricing meant that it would no longer be commercially viable for the Sponsor to launch olmesartan in Australia.




