Dutasteride with tamsulosin hydrochloride, capsule, 500 micrograms – 400 micrograms, Duodart®
Page last updated: 02 March 2011
PDF printable version for Dutasteride with tamsulosin hydrochloride, capsule, 500
micrograms – 400 micrograms, Duodart ® (PDF 189 KB)
Product: Dutasteride with tamsulosin
hydrochloride, capsule, 500 micrograms – 400 micrograms,
Duodart®
Sponsor: GlaxoSmithKline Australia Pty Ltd
Date of PBAC Consideration: November 2010
1. Purpose of Application
The submission sought an Authority required (STREAMLINED) listing
for the treatment of patients with moderate to severe lower urinary
tract symptoms due to benign prostatic hyperplasia (BPH), where
treatment has been initiated by a urologist.
2. Background
The dutasteride with tamsulosin fixed dose combination (FDC) had not previously been
considered by the PBAC.
The PBAC rejected submissions requesting PBS listing for tamsulosin on two occasions
(March and November 2008) on the basis of high and uncertain cost-effectiveness.
At the November 2009 meeting, dutasteride was recommended as an Authority required
(STREAMLINED) listing for the treatment, in combination with an alpha-antagonist,
of lower urinary tract symptoms due to BPH where treatment is initiated by a urologist
on the basis of acceptable cost effectiveness compared with -antagonist alone. Listing
was effective 1 February 2011. A copy of the Public Summary Document (PSD) from this meeting is available.
3. Registration Status
Dutasteride with tamsulosin hydrochloride (Duodart) was TGA
registered on 28 October 2010 for the management of moderate to
severe symptomatic benign prostate hyperplasia.
4. Listing Requested and PBAC’s View
Authority Required (STREAMLINED)
Treatment of moderate to severe lower urinary tract symptoms due to
benign prostatic hyperplasia (BPH) where treatment has been
initiated by a urologist.
For PBAC’s view, see Recommendations and
Reasons.
5. Clinical Place for the Proposed Therapy
BPH is a typically progressive condition associated with worsening
lower urinary tract symptoms such as hesitant and interrupted weak
stream, urgency, leaking or dribbling and a sense of incomplete
voiding leading to more frequent urination, especially at night.
The condition may eventually require surgery. When mild symptoms of
BPH can no longer be managed by ‘watchful waiting’,
current treatment options for patients with moderate to severe
symptoms include treatment with an alpha-antagonist (e.g. prazosin,
tamsulosin, terazosin) and/or a 5 alpha reductase inhibitor (e.g.
finasteride, dutasteride) or surgery.
The submission claimed that the combination product provides an
alternative to taking dutasteride and an alpha blocker as separate
medicines.
6. Comparator
The submission nominated co-administered dutasteride and prazosin
as the main comparator. This was considered appropriate by the
PBAC.
7. Clinical Trials
The submission presented 14 key studies in support of the comparative effectiveness
and comparative toxicity of dutasteride with tamsulosin FDC and co-administered dutasteride
and prazosin.
The indirect analysis of prazosin and tamsulosin modified release (MR) using placebo
as the common comparator included seven trials comparing tamsulosin with placebo (Abrams
1995, Abrams 1997, Chapple 2005, Lepor 1998a, Mohanty 2003, Narayan 1999 and Nordling
2005) and four trials comparing prazosin with placebo (Chapple 1992, Chapple 1990,
Sertcelik 1990 and Steven 1993).
The studies published at the time of the submission are as follows:
| Trial ID / First author | Protocol title / Publication title | Publication citation |
|---|---|---|
| Clinical equivalence of tamsulosin MR with tamsulosin OCAS | ||
| Chapple et al | Tamsulosin oral controlled absorption system (OCAS) in patients with lower urinary tract symptoms suggestive of benign prostatic hyperplasia (LUTS/BPH): Efficacy and tolerability in a placebo and active comparator controlled phase 3a study | Eur Urol Suppl 2005; 4(2): 33–44 |
| Additive benefit of co-administered dutasteride and tamsulosin MR versus tamsulosin MR or dutasteride monotherapy | ||
|
Roehrborn et al Roehrborn et al Becher et al Barkin et al Chung et al Roehrborn et al |
The Effects of Combination Therapy with Dutasteride and Tamsulosin on Clinical Outcomes in Men with Symptomatic Benign Prostatic Hyperplasia: 4-Year Results from the CombAT Study The Influence of Baseline Parameters on Changes in International Prostate Symptom Score with Dutasteride, Tamsulosin, and Combination Therapy among Men with Symptomatic Benign Prostatic Hyperplasia and an Enlarged Prostate: 2-Year Data from the CombAT Study The effects of dutasteride, tamsulosin, and the combination on storage and voiding in men with benign prostatic hyperplasia and prostatic enlargement: 2-year results from the Combination of Avodart ® and Tamsulosin study Effect of dutasteride, tamsulosin and the combination on patient-reported quality of life and treatment satisfaction in men with moderate-to-severe benign prostatic hyperplasia: 2-year data from the CombAT trial Efficacy and safety of dutasteride, tamsulosin and their combination in a subpopulation of the CombAT study: 2 year results in Asian men with moderate-to-severe BPH The effects of dutasteride, tamsulosin and combination therapy on lower urinary tract symptoms in men with benign prostatic hyperplasia and prostatic enlargement: 2-year results from the CombAT study |
European Urology 2010; 57(1):123-131 European Urology 2009; 55(2):461-471 Prostate Cancer and Diseases 2009; 12(4):369-374 BJU International 2009. 103(7):919-926 Prostate Cancer and Prostatic Diseases 2009. 12: 152-159 The Journal of Urology 2008; 179(2):616-621 |
| Indirect comparison (common reference): placebo | ||
| tamsulosin versus placebo | ||
|
Abrams et al Abrams et al Chapple et al Lepor Mohanty et al Narayan et al Nordling |
Tamsulosin, a selective alpha 1c-adrenoceptor antagonist: A randomized, controlled trial in patients with benign prostatic 'obstruction' (symptomatic BPH) A dose-ranging study of the efficacy and safety of tamsulosin, the first prostate-selective alpha1A-adrenoceptor antagonist, in patients with benign prostatic obstruction (symptomatic benign prostatic hyperplasia) Tamsulosin oral controlled absorption system (OCAS) in patients with lower urinary tract symptoms suggestive of benign prostatic hyperplasia (LUTS/BPH): Efficacy and tolerability in a placebo and active comparator controlled phase 3a study Long-term evaluation of tamsulosin in benign prostatic hyperplasia: Placebo-controlled, double-blind extension of phase III trial A double-blind placebo controlled study of tamsulosin in the management of benign prostatic hyperplasia in an Indian population A second phase III multicenter placebo controlled study of 2 dosages of modified release tamsulosin in patients with symptoms of benign prostatic hyperplasia Efficacy and safety of two doses (10 and 15 mg) of alfuzosin or tamsulosin (0.4 mg) once daily for treating symptomatic benign prostatic hyperplasia |
Br J Urol 1995; 76(3): 325–336 Br J Urol 1997; 80(4): 587–596 E ur Urol Suppl 2005; 4(2): 33–44 Urology, 1998; 51(6): 901–906 Ann Coll Surg Hong Kong 2003; 7(3): 88–93 J Urol 1999; 160(5): 1701–1706 BJU Int 2005; 95(7): 1006–1012 |
| prazosin versus placebo | ||
|
Chapple et al Chapple et al Sertcelik Steven et al |
A 12-week placebo-controlled double-blind study of prazosin in the treatment of prostatic obstruction due to benign prostatic hyperplasia A twelve-week placebo-controlled study of prazosin in the treatment of prostatic obstruction Prazosin, a selective alpha 1 receptor blocker in the treatment of benign prostatic hypertrophy. A double-blind placebo-controlled study in Turkey The effect of prazosin on patients with symptoms of benign prostatic hypertrophy |
Br J Urol 1992; 70(3): 285–294 Urol Int 1990; 45(SUPPL. 1): 47–55 Curr Ther Res Clin Exp 1990; 48(6): 1066–1074 Aust Fam Physician 1993; 22(7): 1260–1264 |
8. Results of Trials
The PBAC noted the step-wise approach set out in the submission for examining the comparative effectiveness of the dutasteride/tamsulosin combination with dutasteride/prazosin, which included a 4-step analysis, as follows:
bioequivalence of dutasteride with tamsulosin FDC versus concomitant dutasteride and tamsulosin MR in healthy individuals (trial ARI109882)
clinical equivalence of tamsulosin MR (the formulation of tamsulosin in the FDC) and tamsulosin oral controlled absorption system (OCAS); the formulation currently available in Australia) (Chapple 2005)
additive benefit of co-administered dutasteride and tamsulosin MR with each monotherapy component (CombAT trial – ARI4005, as considered by the PBAC in November 2009, regarding dutasteride)
indirect comparison between prazosin and tamsulosin MR with placebo as the common comparator
The results of the primary outcomes of the trials are presented below.
Bioequivalence of the dutasteride with tamsulosin FDC versus concomitant dutasteride
and tamsulosin MR:
The results of the trial (ARI109882) indicated the bioequivalence of the dutasteride/tamsulosin
FDC and concomitant dosing with separate dutasteride and tamsulosin capsules in either
the fasted or fed state.
Equivalence of tamsulosin MR (the formulation of tamsulosin in the FDC) and tamsulosin
OCAS (the formulation currently available in Australia):
The reduction in International Prostate Symptom Score (IPSS) from baseline was comparable
between tamsulosin MR and tamsulosin OCAS (8.0 versus 7.7 points, p-value not reported
– a comparison of tamsulosin OCAS 0.4 mg and MR 0.4 mg was not an outcome for Chapple
(2005), the publication was assessing whether these formulations were comparably superior
to placebo, p < 0.0001 for both comparisons). The absolute mean change between tamsulosin
MR and placebo and tamsulosin OCAS and placebo (2.0 and 1.7 points for each comparison,
respectively) was comparable between the two tamsulosin formulations, with both formulations
showing superior efficacy compared to placebo, based on a minimally important difference
of ? 1.6 points as defined in the trial design.
Additive benefit analysis of co-administered dutasteride and tamsulosin MR with each
monotherapy component:
After 4 years, combination therapy resulted in a significant reduction in the incidence
of time to first acute urinary retention (AUR) or benign prostatic hyperplasia BPH-related
surgery compared to tamsulosin MR (p < 0.001), and a lower, but non-significant reduction
compared to dutasteride (p=0.18) (4.2% versus 11.9% and 5.2% respectively).
Results for mean change in IPSS from baseline from the CombAT trial are presented
below.
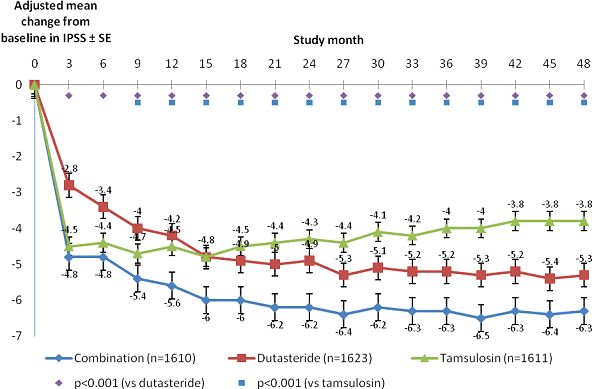
Abbreviations: IPSS: international prostate symptom score; SE: standard error;
The 4-year incidence of BPH clinical progression from the CombAT trial is presented
below.
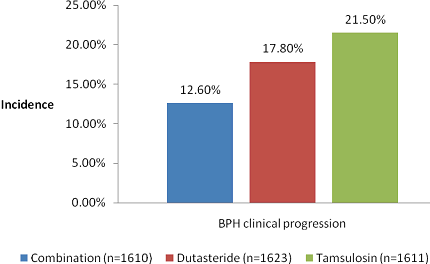
Abbreviations: RRR: reduction in relative risk; CI: confidence interval; BPH: benign
prostatic hyperplasia.
Results for mean adjusted change in Qmax (maximum urine flow) from the CombAT trial
are presented below.
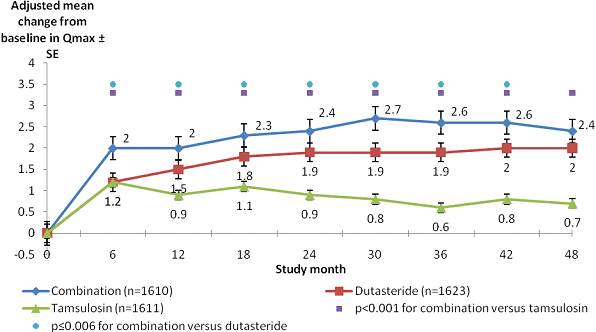
Abbreviations: Qmax: maximum urine flow; SE: standard error.
The PBAC accepted that the dutasteride/tamsulosin FDC was superior to either drug
used as monotherapy.
For PBAC’s view, see Recommendation and Reasons.
Indirect comparison of tamsulosin MR and prazosin using placebo as the common comparator
for the outcome of change in the International Prostate Symptom Score (IPSS):
For the comparison of prazosin and tamsulosin MR, the submission claimed that an improvement
of greater than 2 IPSS points indicated superiority and a difference of no greater
than 1.5 IPSS points indicated non-inferiority. The PBAC has previously agreed that
a 2 point improvement in IPSS score was clinically important.
The indirect comparisons were based on a 2 trials, Mohanty 2003 (tamsulosin MR versus
placebo) and Steven 1993 (prazosin versus placebo). At week 12, the difference in
IPSS score between tamsulosin and prazosin was smaller than at 4 to 8 weeks, totalling
a change of 1.13 points in the favour of tamsulosin. As this difference was less than
1.5 points, tamsulosin was stated to be non-inferior to prazosin at week 12.
The safety profiles of dutasteride monotherapy and tamsulosin monotherapy are well
known and the adverse events (AEs) reported in the key trials indicated that there
was generally little difference in tolerability between the dutasteride with tamsulosin
FDC and the co-administered components.
The combination therapy was associated with an increased relative risk compared to
dutasteride monotherapy (RR: 3.57, 95% CI: 1.17, 10.8) for the pre-defined composite
category of 'cardiac failure' in the CombAT trial. However, the submission claimed
this may be due to chance as no concomitant increase in cardiac events that often
precipitate heart failure (e.g. myocardial infarction, coronary artery disease or
arrhythmias) or an imbalance in AEs normally associated with cardiac failure (e.g.
oedema, dyspnoea or orthopnea) were noted.
9. Clinical Claim
The submission claimed that the dutasteride with tamsulosin FDC is
non-inferior in terms of comparative effectiveness and non-inferior
in terms of comparative safety, compared with co-administered
dutasteride and prazosin.
See Recommendation and Reasons for PBAC’s
view.
10. Economic Analysis
The submission presented a cost minimisation analysis comparing
dutasteride with tamsulosin FDC and co-administered dutasteride and
prazosin. The equi-effective doses were estimated as (i)
dutasteride 0.5 mg and tamsulosin MR 0.4 mg daily for long-term
therapy and (ii) dutasteride 0.5 mg daily and prazosin 2 mg twice
daily for long-term therapy.
11. Estimated PBS Usage and Financial Implications
The likely number of patients per year was estimated in the
submission to be in the range of 100,000 – 200,000 in Year 4,
with savings to the PBS of less than $10 million in Year 4.
12. Recommendation and Reasons
The PBAC recommended the listing of dutasteride with tamsulosin on
a cost minimisation basis compared with dutasteride and prazosin.
The equi-effective doses are (i) dutasteride 0.5 mg and tamsulosin
MR 0.4 mg once daily and (ii) dutasteride 0.5 mg once daily and
prazosin 2 mg twice daily.
The PBAC agreed that the restriction as amended in the
Pre-Sub-Committee response was appropriate. The restriction is the
same as that recommended for dutasteride monotherapy.
The PBAC noted the step-wise approach set out in the submission for
examining the comparative effectiveness of the
dutasteride/tamsulosin combination with dutasteride/prazosin. The
PBAC considered that the totality of the evidence was sufficient to
recommend listing.
The PBAC accepted that dutasteride and tamsulosin MR FDC was
bioequivalent to dutasteride and tamsulosin (MR) taken separately.
The bioequivalence data presented in the submission referred to a
healthy population, and the PBAC accepted that bioavailability
would not be expected to differ in a population with urinary
retention.
On the basis of data from Chapple (2005) presented in the
submission, the PBAC considered that tamsulosin MR and tamsulosin
in an oral controlled absorption system formulation are equivalent.
The data are as follows: symptom improvement as reflected in IPSS
reductions of 8.0 and 7.7 points, respectively, p value not
reported; and absolute mean IPSS improvements over placebo of 2.0
and 1.7 points respectively, where the minimally important clinical
difference was defined as ≥ 1.6 points.
The PBAC accepted that the dutasteride/tamsulosin FDC was superior
to either drug used as monotherapy. The PBAC noted a reduced risk
of acute urinary retention (AUR) and surgery for BPH in patients
taking the FDC; with the FDC, the risk was reduced by 20% compared
to dutasteride monotherapy and 66% compared to tamsulosin
monotherapy. The risk of clinical progression with the FDC (12.6%)
was lower than that with tamsulosin monotherapy (21.5%) and
dutasteride monotherapy (17.8%) (p < 0.001). Improvements in
IPSS from baseline were greater for the FDC (6.3 points) than for
tamsulosin monotherapy (3.8 points) and for dutasteride monotherapy
(5.2 points), and were sustained for 48 months. Improvements in
urinary flow from baseline were greater for the FDC (2.4 mL/sec)
than for tamsulosin monotherapy (0.7 mL/sec), but similar to the
improvement for dutasteride (2.0 mL/sec).
The PBAC agreed that, in the final step of the clinical comparison,
tamsulosin MR
(0.4 mg) had been demonstrated to be non-inferior to prazosin (2 mg
twice daily).
The PBAC noted that, as pointed out by the ESC, only one of the
four trials of prazosin versus placebo reported the outcome of the
change from baseline IPSS (or similar) at 12 weeks. The
comparability of the patients in this prazosin trial reporting this
outcome with those enrolled in the tamsulosin MR trials is
uncertain given the paucity of information regarding eligibility
criteria and baseline characteristics of patients in the prazosin
trial (Steven 1993). However, when the PBAC was making the
recommendation in November 2009 to list dutasteride monotherapy for
use in combination with an alpha-antagonist, members had considered
that the difference in BPH symptom improvement between combination
therapy and alpha-antagonist monotherapy was unlikely to vary
significantly among different alpha-blockers.
The PBAC noted that all but one of the criteria in the fixed
combination products guidelines had been met. The criterion that
was not met was that the individual components of a FDC should be
listed on the PBS as monotherapy. On this occasion, the recommended
restriction for dutasteride specifies use in combination with an
alpha-antagonist. Tamsulosin may be a preferred option for patients
with BPH and does not require dose titration. The PBAC thus
considered that the fact that tamsulosin was not PBS-listed should
not stand in the way of listing.
The PBAC recommended the Safety Net 20 Day rule should apply.
The PBAC recommended that dutasteride with tamsulosin fixed dose
combination is suitable for inclusion in the PBS medicines for
prescribing by nurse practitioners, as continuing therapy
only.
Recommendation:
DUTASTERIDE 0.5 mg with TAMSULOSIN HYDROCHLORIDE 0.4 mg,
capsule
Restriction: Authority Required (STREAMLINED)
Treatment of lower urinary tract symptoms due to benign prostatic hyperplasia where treatment has been initiated by a urologist.
NOTE:
Continuing therapy only:
For prescribing by nurse practitioners as continuing therapy only, where the treatment of, and prescribing of medicine for, a patient has been initiated by a medical practitioner. Further information can be found in the Explanatory Notes for Nurse Practitioners.
Maximum quantity: 30
Repeats: 5
13. Context for Decision
The PBAC helps decide whether and, if so, how medicines should be
subsidised in Australia. It considers submissions in this context.
A PBAC decision not to recommend listing or not to recommend
changing a listing does not represent a final PBAC view about the
merits of the medicine. A company can resubmit to the PBAC or seek
independent review of the PBAC decision.
14. Sponsor’s Comment
The sponsor is happy with the outcome.




