Australian Statistics on Medicines 2011
Page last updated: 28 January 2014
Australian Statistics on Medicines 2011 (PDF 5.2 MB)
Table 1 (Excel 357 KB)
Table 2 (Excel 244 KB)
Acknowledgments
Prepared by Vanna Mabbott, Maxine Robinson and Alicia Segrave of the Pharmaceutical Benefits Division.
We would like to thank the following people for their help in the access and provision of data and information used in this report:
- The World Health Organization Collaborating Centre for Drug Statistics Methodology.
- Kavita Verma, Pharmaceutical Access, Department of Health.
- Christopher Parker, Pharmaceutical Access, Department of Health.
- Katherine Gray, Advisory Committee on the Safety of Medicines, Therapeutic Goods Administration.
- Rebecca Bennetts, Australian Institute of Health and Welfare.
Australian Statistics on Medicine 2011
Print ISBN: 978-1-74186-083-2
Online ISBN: 978-1-74186-084-9
Publications approval number: 10534
Copyright Statements:
Paper-based publications
© Commonwealth of Australia 2013
This work is copyright. You may reproduce the whole or part of this work in unaltered form for your own personal use or, if you are part of an organisation, for internal use within your organisation, but only if you or your organisation do not use the reproduction for any commercial purpose and retain this copyright notice and all disclaimer notices as part of that reproduction. Apart from rights to use as permitted by the Copyright Act 1968 or allowed by this copyright notice, all other rights are reserved and you are not allowed to reproduce the whole or any part of this work in any way (electronic or otherwise) without first being given the specific written permission from the Commonwealth to do so. Requests and inquiries concerning reproduction and rights are to be sent to the Online, Services and External Relations Branch, Department of Health, GPO Box 9848, Canberra ACT 2601, or via e-mail to copyright@health.gov.au.
Internet sites
© Commonwealth of Australia 2013
This work is copyright. You may download, display, print and reproduce the whole or part of this work in unaltered form for your own personal use or, if you are part of an organisation, for internal use within your organisation, but only if you or your organisation do not use the reproduction for any commercial purpose and retain this copyright notice and all disclaimer notices as part of that reproduction. Apart from rights to use as permitted by the Copyright Act 1968 or allowed by this copyright notice, all other rights are reserved and you are not allowed to reproduce the whole or any part of this work in any way (electronic or otherwise) without first being given the specific written permission from the Commonwealth to do so. Requests and inquiries concerning reproduction and rights are to be sent to the Online, Services and External Relations Branch, Department of Health, GPO Box 9848, Canberra ACT 2601, or via e-mail to copyright@health.gov.au.
FOREWORD
It is my great pleasure to introduce the 17th Edition of the Australian Statistics on Medicines. This publication provides an extensive and extremely valuable set of statistics on the use of prescription medicines in Australia from 1991 to 2011. Data in this edition were obtained from the Pharmaceutical Benefits Scheme, the Repatriation Pharmaceutical Benefits Scheme and an ongoing survey of a representative sample of community pharmacies.
Knowledge about the usage, cost and trends over time in prescription medications is clearly relevant for all four aspects of the National Medicines Policy: access to medicines; quality, safety and efficacy of medicines, quality use of medicines; and a responsible and viable medicines industry. These data are routinely scrutinized by the Drug Utilisation Sub-Committee (DUSC) which has provided advice to the Pharmaceutical Benefits Advisory Committee and other stakeholders within the National Medicines Policy Framework about the use (predicted and actual) of medicines in Australia since 1988. This advice is critical with the number, usage, cost and complexity of prescription medicines increasing with an ageing population and ongoing development of new and innovative medicines. Data from the Australian Statistics on Medicines and advice from DUSC provide an integral contribution to the monitoring and quality assurance of the Pharmaceutical Benefits Scheme, prescribers and the medicines industry.
This edition of the Australian Statistics on Medicines was prepared by staff of the Pharmaceutical Benefits Division. I acknowledge their skill and expertise and thank them for their extraordinary commitment and hard work. The Australian Statistics on Medicines has a critical role in our health care system and provides information that ultimately improves the health of Australians.
Geoff McColl
FRACP PhD
Chair, Drug Utilisation Sub-Committee
Contents
- FOREWORD
- INTRODUCTION
- INFORMATION ON THE AUSTRALIAN STATISTICS ON MEDICINES
- Overview
- Pharmaceutical Benefits Advisory Committee
- Drug Utilisation Sub-Committee
- National Medicines Policy
- Drug Classification
- Measurement Unit
- Department of Human Services processing
- Pharmacy Guild Survey data
- Combined database
- ADVERSE EVENT REPORTING IN AUSTRALIA IN 2011
- THE HIGHLY SPECIALISED DRUGS PROGRAMME
- SUPPLY OF PHARMACEUTICAL BENEFITS TO REMOTE AREA ABORIGINAL HEALTH SERVICES UNDER SECTION 100 OF THE NATIONAL HEALTH ACT
- HEALTH EXPENDITURE TRENDS
- DRUG UTILISATION TRENDS
- TABLES IN THE AUSTRALIAN STATISTICS ON MEDICINES
- CAVEATS
- GLOSSARY OF TERMS
- ATC & DDD Additions and Alterations
- TABLE 1: 2011 COMMUNITY PRESCRIPTION NUMBERS, TOGETHER WITH GOVERNMENT AND PATIENT COSTS FOR PBS LISTED DRUGS
- TABLE 2: COMMUNITY PRESCRIPTION DRUG USE, IN DDD/1000/DAY, FOR 2009 TO 2011
- ATC INDEX 2012
- TABLES AND FIGURES
- List of Tables
- Table A: Highly Specialised Drugs—National Usage and Patient Report in Public Hospitals for the period January 2011 to December 2011
- Table B (i): Total expenditure on pharmaceuticals and other medical non-durables as % total expenditure on health, TEH
- Table B (ii): Total expenditure on pharmaceuticals & other medical non-durables per capita, US$ purchasing power parity
- Table B (iii): Total expenditure on health as % gross domestic product
- Tables C: Prescription numbers by ATC groups
- Table C (i): Subsidised prescriptions (PBS/RPBS)
- Table C (ii): Estimated non-subsidised prescriptions (Survey)
- Table D: Top 10 drugs by defined daily dose/thousand population/day, 2011 (including the contribution of constituents of combination products)
- Table E:Top 10 drugs by prescription counts, 2011
- Table F: Top 10 PBS/RPBS drugs by total cost to Australia, 2011
- Table G: Nurse Practitioner—Top 10 PBS/RPBS drugs by prescription counts, 2011
- Table H: Nurse Practitioner—Top 10 PBS/RPBS drugs prescribed by total cost, 2011
- Table I: Optometrist—Top 10 PBS/RPBS drugs by prescription counts, 2011
- Table J: Optometrist—Top 10 PBS/RPBS drugs prescribed by total cost, 2011
- List of Figures
INTRODUCTION
The data contained in the 2011 Australian Statistics on Medicines are drawn from two sources. The first is the Department of Human Services records of prescriptions submitted for payment of a subsidy under the Pharmaceutical Benefits Scheme (PBS) and Repatriation Pharmaceutical Benefits Scheme (RPBS). The second is a survey of a representative sample of community pharmacies, which provides an estimate of the non-subsidised use of prescription medicines in the Australian community. The usage of prescription medicines dispensed to in-patients in public hospitals is not available in this report. The usage of subsidised PBS/RPBS prescription medicines to out-patients and discharged patients in five states of Australia and one territory are included. It is planned that all out-patients and discharged patients will receive PBS subsidised prescriptions in the future. The units of measurement are the prescription and the defined daily dose per 1000 population per day (DDD/1000 population/day). The defined daily dose is established by the World Health Organization Collaborating Centre (WHOCC) for Drug Statistics Methodology on the basis of the assumed average dose per day of the drug, used for its main indication by adults. The drugs presented in this publication are arranged using the Anatomical Therapeutic Chemical (ATC) classification system. For more detail on this classification and the unit of measurement, please read the chapter ‘Information on the Australian Statistics on Medicines’. The data are presented in two major tables. Table 1 includes 2011 community (i.e. subsidised and non-subsidised) prescription numbers. These figures are presented together with the government and patient costs for drugs PBS listed and subsidised by the Australian Government only. Cost information on the dispensing of drugs not listed on the PBS and drugs that are PBS-listed but for which no subsidy is claimed from the Australian Government is not available for the period of this report. Collection of under general co-payment PBS volume data commenced on 1 April 2012, and will inform future reports thereafter. Table 2 includes community prescription drug use, in DDDs/1000 population/day, for the years 2009, 2010 and 2011. Table 2 reports the DDDs for each drug, reporting the use in monocomponent (‘plain’) and in fixed dose combination formulations.
INFORMATION ON THE AUSTRALIAN STATISTICS ON MEDICINES
Overview
The development, monitoring and promotion of rational and cost-effective use of medications in society are dependent on accurate information on patterns of drug prescription and use. Where use is considered to be inappropriate, drug utilisation data can monitor the impact of educational or regulatory interventions, and can guide the interpretation of pharmacoeconomic analysis1.
In Australia, community prescriptions (i.e. non-public hospital) are dispensed either as private prescriptions, funded by the patient or private health insurer, or under one of two Government subsidisation schemes—the Pharmaceutical Benefits Scheme (PBS) and the Repatriation Pharmaceutical Benefits Scheme (RPBS). These schemes were established to provide the general community (PBS) and returned servicemen and women (RPBS) with access to necessary medicinal products which are affordable, available and of acceptable standards. Since 2002 prescriptions for an increasing number of public hospital outpatients and many medicinal products supplied at discharge for in-patients have been included in the dataset. In 2011, the RPBS was 6% of the size of the PBS and a large majority, approximately 93%, of RPBS prescriptions involved PBS listed drugs.
In Australia, a new medicinal drug must gain approval for supply in accordance with the requirements of the Therapeutic Goods Act 1989. Approval is also required to extend the indications of an established drug. Applications are dealt with by the Therapeutic Goods Administration (TGA) and, for prescription drugs, advice is sought from an expert committee. From 1963 to 2009 this advice was provided by the Australian Drug Evaluation Committee (ADEC). ADEC was replaced in January 2010 with the Advisory Committee on Prescription Medicines (ACPM).
Once a prescription drug is approved for marketing, the company concerned usually applies to have the drug listed on the PBS. This is the national scheme available to the Australian community for subsidising the cost of pharmaceuticals. The subsidised cost, particularly for newer drugs, reduces consumers’ out of pocket expenses therefore many companies seek to have the drug listed on the scheme to facilitate viable marketing.
The Pharmaceutical Benefits Advisory Committee (PBAC) makes recommendations to the Australian Government about which drugs should be listed on the PBS. Pre-market evaluation addresses the issues of quality, safety and efficacy, whereas the PBAC considers effectiveness and cost-effectiveness of the product relative to alternatives, as well as the overall cost to the Government. Once the PBAC has recommended a drug for listing on the PBS, the Pharmaceutical Benefits Pricing Authority (PBPA) negotiates the price with the sponsor company. The PBPA consists of government, industry and consumer representatives. After agreement is reached, the Australian Government considers the advice of both the PBAC and the PBPA and makes a decision on whether the drug will be listed on the PBS.
Under the PBS, patient contributions towards medication costs at pharmacies are capped. In 2011, general patients paid the cost of a prescription up to a maximum of $34.20. Pensioner and concession patients paid $5.60 per prescription.
In addition, there is a Safety Net Scheme to protect people with high medication needs. In 2011, once general patients and/or their immediate family incurred $1,317.20 of PBS expenditure (indexed), PBS/RPBS prescriptions for the remainder of the calendar year cost only $5.60 per prescription. Once pensioners and other concession card holders reached the concession safety net threshold of $336 expenditure (indexed), they received all remaining prescriptions in 2011 free of charge.
It is important to note that patients may be required to pay a surcharge if a doctor prescribes a more expensive brand of an item, when there are cheaper, equivalent brands of that item listed on the PBS.
As the general patient co-payment rises, the dispensed price of many cheaper medical products fall under this level. In such cases the patient pays the full price and no claim for payment was transmitted under the PBS for the period of this report. In 2011, under co-payment general prescriptions represented around 17.9% of all community prescriptions. There are also many drugs that are not listed on the PBS or RPBS and are available only on private prescription, with the patient paying the full cost. Private prescriptions represented 7.2% of community prescriptions in 2011.
Pharmaceutical Benefits Advisory Committee
The Pharmaceutical Benefits Advisory Committee (PBAC) is an independent statutory body established on 12 May 1954, under section 100A of the National Health Act 1953. The role of PBAC is to make recommendations and give advice to the Minister about which drugs and medicinal preparations should be made available as pharmaceutical benefits. No new drug may be made available as a pharmaceutical benefit unless recommended by the PBAC.
The PBAC is required by the Act to consider the effectiveness and cost of a proposed benefit compared to alternative therapies. In making its recommendations, the PBAC, on the basis of expected community usage, recommends maximum quantities and repeats, and may also recommend restrictions as to the indications where PBS subsidy is available. When recommending listings, the PBAC provides advice to the PBPA regarding comparison with alternatives or their cost effectiveness.
Drug Utilisation Sub-Committee
In 1988, the PBAC convened the Drug Utilisation Sub-Committee (DUSC) to assist it in making recommendations for listings on the PBS. Its terms of reference are:
- To develop and advise on the mechanisms for the collection, analysis and interpretation of comprehensive data on utilisation of medicines in Australia.
- To advise PBAC on changes in patterns of utilisation of medicines as a consequence of changes in their availability or subsidy restrictions and to review the utilisation of medicines, including but not restricted to expenditure impacts within the Pharmaceutical Benefits Scheme (PBS).
- To advise stakeholders within the National Medicines Policy framework on the interpretation of patterns of utilisation of medicines, including by placing the results of the data in the context of the limitations of the data.
- To identify potential problems and benefits related to patterns of utilisation of medicines.
- To evaluate policy and other interventions related to the use of medicines.
- To facilitate and promote the dissemination of information on utilisation of medicines.
- To conduct international comparisons of utilisation of medicines by interaction with
appropriate
international bodies.
National Medicines Policy
The National Medicines Policy (NMP) is a broad framework that aims to improve health outcomes for all Australian’s through access to and appropriate use of medicines.
The overall aim of the NMP is to meet medication and related service needs, so that both optimal health outcomes and economic objectives are achieved. In this context “medicines” means prescription, non-prescription and complementary healthcare products.
The NMP has four central objectives:
- timely access to the medicines that Australian’s need, at a cost individuals and the community can afford;
- medicines meeting appropriate standards of quality, safety and efficacy;
- quality use of medicines;
- and maintaining a responsible and viable medicines industry.
Further information is available on the National Medicines Policy web page.
Drug Classification
The DUSC and the Department of Health have adopted the Anatomical Therapeutic Chemical (ATC) code as recommended by the World Health Organization (WHO). It has been a goal of WHO to have an internationally accepted classification for presenting and comparing drug usage data. In 1982, the WHO Collaborating Centre for Drug Statistics Methodology (WHOCC), located in Norway, was established as a central body responsible for coordinating ATC use.
The ATC code itself is a seven digit alphanumeric code, structured in five levels, that classifies drugs according to their site of action and therapeutic and chemical characteristics.
The first level of the code is the anatomical main group. There are 14 anatomical main groups. The second and third levels are for the therapeutic subgroup and pharmacological subgroup, respectively, with a fourth level being either a chemical or therapeutic subgroup. The fifth level is the actual chemical substance.
The five levels thus are:
- anatomical main group
- pharmacological/therapeutic subgroup
- chemical/pharmacological or therapeutic subgroup
- chemical/pharmacological or therapeutic subgroup
- chemical substance (generic drug name)
For example, risperidone has the following code:N 05 A X 08.
| N denotes | Nervous system |
| 05 | Psycholeptics |
| A | Antipsychotics |
| X | Other antipsychotics |
| 08 | Risperidone |
ATC system main groups:
The 14 anatomical main groups of the ATC code are:
| A. | Alimentary tract and metabolism |
| B. | Blood and blood forming organs |
| C. | Cardiovascular system |
| D. | Dermatologicals |
| G. | Genitourinary system and sex hormones |
| H. | Systemic hormonal preparations, excluding sex hormones and insulins |
| J. | Anti-infectives for systemic use |
| L. | Antineoplastic and immunomodulating agents |
| M. | Musculo-skeletal system |
| N. | Nervous system |
| P. | Antiparasitic products, insecticides and repellents |
| R. | Respiratory system |
| S. | Sensory organs |
| V. | Various |
Although the ATC code extends to the generic drug level, it does not identify dosage forms, pack sizes, strengths or brand names.
The WHOCC, together with the Nordic Council on Medicines, undertakes regular revisions of the ATC system. They receive expert advice from an advisory board and an established procedure exists to manage requests for new classifications and to regularly review the current structure. Changes implemented in 2012 are included in the Anatomical Therapeutic Chemical Index (ATC) & Defined Daily Dose (DDD) additions and alterations section in this publication.
Measurement Unit
The international unit of drug utilisation adopted by the DUSC to accompany this coding system is the defined daily doses, per thousand of the population, per day (DDDs/1000/day). The defined daily dose is established by the Nordic Council on Medicines and the WHO Drug Utilisation Research Group on the basis of the assumed average dose per day of the drug, when used for its main indication by adults3.
Use of DDDs allows for comparisons of drug utilisation independent of differences in price, preparation and quantity per prescription. It also allows comparison of the use of drugs in different therapeutic groups, and between regions and countries. Expressing drug use in DDDs/1000/day allows the aggregation of data for those drugs which have differing daily doses. The DDD, however, is only a technical unit of use and does not necessarily reflect the recommended or average prescribed dose in Australia.
The DDDs/1000/day figure is calculated from prescription data in the following way:
|
N x M x Q x 1000 |
|
DDD x P x D |
Where:
| N | is the number of prescriptions dispensed in the year |
| M | is the drug mass in each unit (tablet, capsule, injection, pack etc.) (e.g. milligrams or grams, expressed in the same unit as DDD) |
| Q | is the average dispensed quantity (ie. number of units)per prescription |
|
P |
is the midyear Australian population for the year of data collection (see Australian Bureau of Statistics website for population figures used in this edition. |
| D | is the number of days in the year. |
The DDDs/1000/day can be calculated over other time periods such as monthly or quarterly.
For PBS items, the mass amount (M) is the amount of active drug contained in an individual dose unit e.g. tablet, capsule, suppository etc. Non-PBS items are estimated from the Pharmacy Guild survey. The data from the survey does not include information on the quantity supplied per prescription, therefore the mass amount for nonsubsidised items is the total amount of active drug contained in the pack.
For prescriptions forwarded for subsidy, the average quantity dispensed (Q), is available from Department of Human Services data. For prescriptions that are priced under the general co-payment, quantity is assumed to be the average quantity of the subsidised prescriptions for that drug (i.e. as concession, safety net and Veterans’ Affairs (Repatriation) prescriptions). For private prescriptions, the quantity dispensed is assumed to be the retail pack size.
For a chronically administered drug, the DDDs/1000/day figure indicates how many people, per 1000 of the population, may, in theory, have received a standard dose (as defined by the DDD) daily.
For drugs used intermittently, for example antiinfectives, usage expressed in DDDs/1000 /day may similarly give a rough estimate of the average proportion of the population using these drugs every day. To estimate the number of patients treated during the year supplementary information, such as the average duration of treatment, is necessary3.
The ATC/DDD methodology has a number of limitations. All drugs dispensed are not necessarily consumed and the DDDs/1000/day is calculated for the total population, while drug use may be concentrated in certain age groups or a particular sex.
It is difficult to assign a DDD, and on occasions an ATC code, to some preparations that have multiple active ingredients. For some drug groups, such as the dermatological and antineoplastic drugs, highly individualised use and wide dose ranges, as well as the experimental nature of some of the therapy, make it difficult to define a daily dose. Consequently, there may be a delay between the marketing of a drug and the availability of an ATC code and its associated DDD.
Generally agreed indications for use of a drug may be re-evaluated in light of experience with adverse reactions and other pharmacological effects. Drugs may have multiple indications and it may be difficult to determine a preparation’s use. Also, the DDD is based on overseas experience and may not reflect the prescribed adult dose in Australia.
As more medicinal products are listed on the PBS in formulations of two or more combinations the DUSC has considered that it is important to record the contribution, in terms of DDDs, of each constituent where appropriate. Therefore additional information on the contribution of the constituents of combination pharmaceutical items in addition to single component items will be reported in table 2.
Department of Human Services processing
In 1990, the processing of prescriptions submitted for payment of a subsidy under the PBS/RPBS was taken over by the Health Insurance Commission, now the Department of Human Services. Daily data transmissions, containing prescription records that do not allow the identification of an individual patient, are provided by the Department of Human Services to the Department of Health for summarisation.
Pharmacy Guild Survey data
Since 1989, DUSC has commissioned the Pharmacy Guild of Australia to conduct an annual survey to estimate the prescription volumes for drugs in the nonsubsidised categories i.e. private prescriptions and PBS prescriptions priced under the general patient co-payment. Total dispensing information is collected each month from pharmacies that are members of the Pharmacy Guild. The sample increased in 2007 from 150 to 370.
Amfac, a major pharmacy computer software supplier, was commissioned to administer the collection of the data in 1988. Under the joint direction of DUSC and the Guild, Amfac contracted a firm of statisticians, specialising in survey design and analysis, to design a stratified random sample, using the Guild membership, which represents approximately 80% of pharmacies in Australia, as the population base. In 1993, the survey sample was reviewed and augmented with the assistance of the Statistical Services Section within the Department. A review of the representativeness, sample size, design and risks for the survey was carried out by the Australian Bureau of Statistics (ABS) in February 2002. It found a small relative standard error, for the sample size of 150 pharmacies, of 4.3%.
In the original form of the survey, dispensing records from the participating pharmacies were sent to Amfac’s Canberra premises. Several hundred diskettes were summarised by drug code and category. A single disk was then forwarded to the Department. Details of the dispensing of individual participating pharmacies are not available on these data.
The Survey data was not supplied by the Guild from September 1999 to February 2001. When monthly data collection recommenced in February 2001, retrospective data was retrieved using an internet connection between each participating pharmacy and a software provider. A total of 142 pharmacies participated in this data collection.
Following reinstatement of the Survey, data is now transmitted electronically from the participating pharmacy dispensaries to the software providers. Data is forwarded to the Guild and then to DUSC. The data continues to be de-identified with respect to individual pharmacies and individual patient prescriptions.
The pharmacies in the survey are selected to be representative of the population of operational pharmacies with regard to PBS dispensing volume and geographical location, and are similarly stratified. In order to compare and then extrapolate the survey quantities to estimates of use in Australia, the subsidised PBS prescription data supplied by all pharmacies in Australia are stratified into the same four equal dispensing volume ranges based on their annual average PBS dispensing from the previous year. A weighting factor is calculated for each PBS and Amfac item code by comparing the number of pharmacies and PBS prescriptions in the survey with the total number of pharmacies and PBS prescriptions in Australia for each stratum. Volumes of non-subsidised drug use are calculated by multiplying the survey estimate by the weighting factor, which is assumed to apply equally to the subsidised and nonsubsidised prescription volumes.
Combined database
A Departmental database combines the prescription estimates for the non-subsidised sector, under general co-payment and private prescriptions, based on the survey with the actual counts of those prescription categories submitted to the Department of Human Services for payment of a PBS/RPBS subsidy by the Government. A number of other Government subsidised programs and PBS programs subsidised through alternative supply arrangements are not included in the DUSC database. This includes the use of highly specialised drugs available for outpatients through public hospital pharmacies under Section 100 Highly Specialised Drugs Program of the National Health Act 1953, supply of drugs to remote area Aboriginal Health Services, Opioid Dependence Scheme, IVF/ GIFT Treatment and Botulinum Toxin Program. Subsidised programs operated by State Health Departments, including supply of drugs to public hospital in-patients, and the Herceptin Program for metastatic breast cancer are also excluded from the DUSC database. The combined dataset does contain information on highly specialised drugs and in-patient drugs provided through private hospitals as this information is collected by the Department of Human Services. Following a range of pharmaceutical reforms a greater number of s100 Highly Specialised Drugs, and outpatient or discharge drugs supplied through public hospital will be included in the DUSC database in the future.
The advantages of the expanded database can be illustrated by using an example involving the drug sotalol hydrochloride. Previously, sotalol hydrochloridehad a price per prescription, as a general benefit, above the patient co-payment and, as a consequence, a majority of community use was captured on the PBS/RPBS claims database. In 2008, a reduction in drug price and a small increase in the general patient co-payment in line with the Consumer Price Index (CPI) contributed to the price of sotalol hydrochloride falling below the general patient co-payment. Changes in the capturing of sotalol hydrochloride data from the Department of Human Services claims database to the Pharmacy Guild Survey database can be seen in figure A. The combined database therefore enables the continuation of utilisation estimates for drugs not subsidised through the PBS/RPBS and provides a more comprehensive outlook on drug use within Australia.
Figure A shows the time trends for dispensing of sotalol hydrochloride, by the subsidised and non-subsidised components.
Figure A: Community utilisation of sotalol hydrochloride
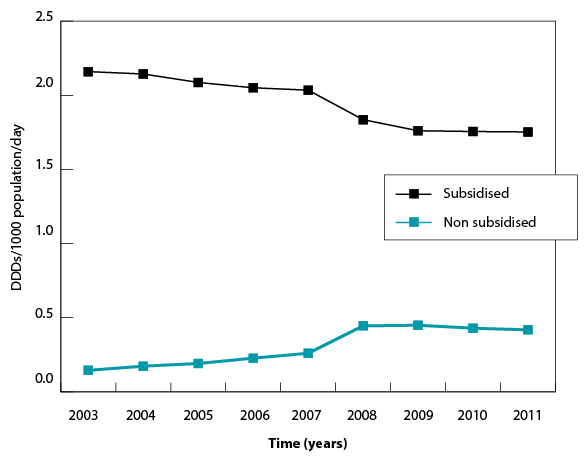
Quantities within the PBS Schedule are designed to provide a normal course of treatment for acute conditions, and a month’s treatment at usual doses for chronic conditions.
A pattern involving PBS drug utilisation that shows a higher level of usage leading up to the end of a year has been previously reported4. This peak is due to the safety net provisions introduced into the PBS in November 1986. These provisions were introduced to financially support patients with multiple medical conditions who genuinely need a number of medicines. Once the out-of-pocket threshold safety net level is reached, prescriptions on the scheme are either free, or available at a greatly reduced co-payment amount. The safety net period is the calendar year, and the highs and lows are due to stockpiling of medication once the safety net level is reached.
The stockpiling of medication has public health, waste and cost implications. Large quantities of potent medicines in the home can be a hazard for other family members, may exceed their expiry date, and has the potential for patient confusion if the dosage or the need for a particular medication is subsequently reviewed by the doctor during this period.
On 1 November 1994 the National Health (Pharmaceutical Benefits) Regulations were amended to increase the period for redispensing chronically used drugs (i.e. those with 5 or more repeats) to a period of no less than 20 days. Exceptions are determined by the PBAC and include eye drops, which tend to be used at a higher rate than other medications. The redispensing period here was amended to four rather than the previous three days.
The pharmacist has the discretion to supply earlier than the statutory period if the circumstances warrant e.g. medicine lost or prescribed dosage requires more frequent dispensing of repeats.
ADVERSE EVENT REPORTING IN AUSTRALIA IN 2011
The Therapeutic Goods Administration (TGA) is responsible for regulating medicines in Australia, including monitoring the ongoing safety of medicines once they have been included on the Australian Register of Therapeutic Goods (ARTG).
The TGA’s reporting system for adverse events began in the late 1960’s with the computerised database dating back to November 1972. At the end of 2011 there were approximately 247,000 reports of suspected adverse events in the database.
Figure B: Origin of adverse event reports received by the TGA (2006-2011)
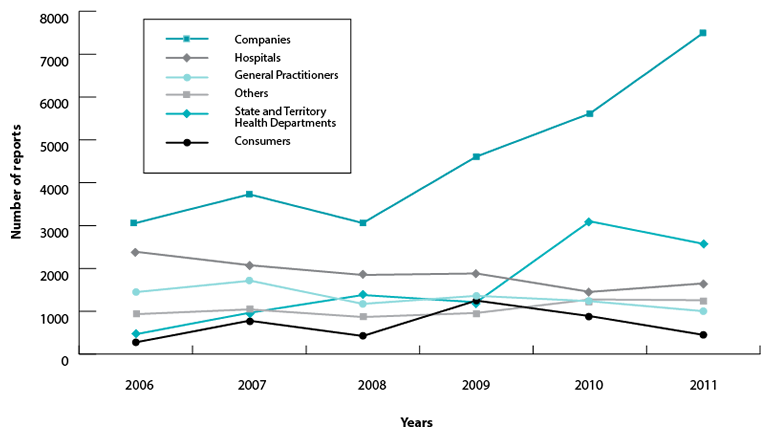
Note: A review of the quality control process had an impact on the searchable part of the Adverse Drug Reaction Database, causing a change in the number of events from data published in the Adverse Drug Reaction Reporting in Australia in 2010.
In 2011 the TGA received approximately 14,400 reports with 52% from pharmaceutical companies, 12% from hospitals, 7% from General Practitioners (GPs), 18% from State and Territory Health Departments and 3% from consumers. The sources for other reports (8%) include community pharmacists and specialists (Figure B). On average, in 2011, the TGA received 1,200 reports per month. Health professionals are encouraged to report suspected adverse events directly to the TGA rather than through the sponsor or manufacturer to simplify communication.
The spike in the number of adverse events reported by companies in 2011 is attributed to a number of factors, including a greater number of drugs on the Australian Register of Therapeutic Goods (ARTG); and the possibility that GPs are reporting adverse events via sponsors, which would also account for the slight drop in the reporting rates for GPs. The increases observed in 2009 and 2010, particularly from State and Territory Health Departments and companies, is largely due to reports of adverse events following vaccination with the pandemic (H1N1) influenza vaccine in 2009 and the seasonal trivalent influenza vaccines in 2010.
Processing and use of adverse event reports
Reports are assessed by the Office of Product Review (OPR) within the TGA. This involves checking the report for the presence of ‘minimum’ details, i.e. an individual patient, an adverse event, at least one (suspected) drug, and, preferably, an identifiable reporting health professional. The specific reaction terms are identified along with the suspected, interacting or ‘other’ drugs and these are entered into the database.
The TGA applies a causality rating for the reaction(s) and in some cases requests further clinical or laboratory information from the reporter to allow causality to be assessed. Medical officers review serious reports and staff in OPR regularly analyse reporting data to identify potential safety signals.
Reports are forwarded to the Uppsala Monitoring Centre in Sweden which administers the WHO Collaborating Centre for International Drug Monitoring. This global database began in 1968 as a pilot program involving 10 nations, including Australia, and now receives reports from over 80 nations with over 5 million reports in the database.
Reporting adverse events
The TGA encourages the reporting of all suspected adverse events to medicines available in Australia, including prescription medicines, vaccines, over the counter and complementary medicines. The reporting of seemingly insignificant or common adverse events can contribute to the TGA’s investigation of a potential safety signal.
The TGA particularly requests reports of:
- all suspected reactions to new medicines
- all suspected medicines interactions
- unexpected reactions (i.e. those reactions that are not described in the Product Information)
- suspected reactions causing:
- death
- admission to hospital or prolongation of hospitalisation
- increased investigations or treatment
- birth defects
Reports of suspected adverse events can be made:
- online at www.tga.gov.au by following the link to ‘Report a Problem’. Health professionals can also submit a report directly from MIMS online.
- using a ‘Blue Card’ available from the TGA’s Office of Product Review (1800 044 114 or adr.reports@tga.gov.au) or downloaded from the TGA website at www.tga.gov.au/safety/problem-medicines-forms-bluecard.htm.
Expert advisory committee
The Advisory Committee on the Safety of Medicines (ACSOM) was established in January 2010 to provide expert advice to the TGA about safety issues under investigation and the appropriateness of Risk Management Plans (RMPs). RMPs outline sponsors’ plans to monitor and communicate risks, and are evaluated as part of the registration process for new medicines. RMPs accompany applications for registration of high risk medicines, such as new chemical entities. RMPs characterise and pro-actively manage risks relating to a medicine over its entire life cycle. ACSOM also provides advice to the TGA on other matters related to pharmacovigilance, including the detection, assessment, understanding and prevention of adverse events.
Medicines Safety Update
The Medicines Safety Update is published six times a year in Australian Prescriber, and also on the TGA website. The Medicines Safety Update replaced the Australian Adverse Drug Reactions Bulletin in 2010.
The following articles were published in Medicines Safety Update in 2011:
- Clozapine and severe constipation
- Drug interaction between tamoxifen and antidepressants
- Methysergide and retroperitoneal fibrosis
- Thank you for your reports
- Suspected adverse reactions to vaccines:a reminder to report
- Drug-induced hyponatraemia
- Rotavirus vaccination and risk of intussusception: investigation of a possible safety signal
- Coversyl and Coumadin: new packaging to reduce potential for dispensing errors
- Risk of hypomagnesaemia with proton pump inhibitors
- Use of 2011 seasonal influenza vaccines in children
- Investigation of Prevenar and deaths in children in Japan: what does it mean for Australia?
- Finding information about adverse reaction reporting on the new TGA website
- Medicine recalls in Australia
- Cramps, quinine and thrombocytopenia
- Venlafaxine and stress cardiomyopathy
- In utero antipsychotic exposure and neonatal extrapyramidal and withdrawal adverse effects
- Prescribing medicines in pregnancy - new TGA database
- Oral contraceptives containing drospirenone (Yaz and Yasmin) and venous thromboembolism
- Pioglitazone and risk of bladder cancer
- Modafinil (Modavigil) - safety update
- Subscribe to TGA email alerts
- Proton Pump Inhibitors (PPIs) and acute interstitial nephritis
- Dabigatran (Pradaxa) and the risk of bleeding
- Reducing the risk of myopathy and rhabdomyolysis with simvastatin - new dosage recommendation
Transparency
Recent reviews of the functions and activities of the TGA have identified a need to ensure the public is aware of the activities undertaken by the TGA to monitor the safety of therapeutic products available to the community. In December 2011 the Government released TGA reforms: a blueprint for TGA’s future (the Blueprint) which included plans to improve the Australian community’s understanding of the TGA’s regulatory processes and decisions and enhance public trust in the safety and quality of therapeutic goods.
The Blueprint consists of a range of interconnected projects the TGA will undertake to achieve more effective and transparent regulation of medicines, meet emerging challenges in the regulation of therapeutic goods and provide more timely information about the goods it regulates.
The TGA applies a risk management approach to ensure therapeutic goods supplied in Australia meet acceptable standards of quality, safety and efficacy. Once a therapeutic product is approved, the TGA continues to monitor the product in the market through therapeutic product vigilance activities.
Product vigilance
The aim of therapeutic product vigilance is continually to monitor and evaluate the safety and efficacy (performance) profile of therapeutic products and to manage any risks associated with individual products over the life cycle of a product. The TGA’s therapeutic product vigilance framework is available on the TGA website at www.tga.gov.au/safety/tga-therapeutic-product-vigilance.htm.
The maintenance and improvement of health and safety is a shared responsibility. In addition to government and regulated industry, health professionals, consumers and their respective associations play an important role in reporting therapeutic product safety related issues.
Sponsors have the primary responsibility for the safety of any therapeutic products they import into, supply in or export from Australia. Sponsors must comply with legislative requirements for therapeutic product vigilance under the Therapeutic Goods Act 1989 and there are applicable offences and penalties under the Act for not complying. The legislative requirements for therapeutic product vigilance vary depending on the type of therapeutic good.
The TGA maintains up-to-date safety information on therapeutic products that is communicated through a variety of means to health professionals and consumers. The TGA is committed to advancing public health through the market authorisation of beneficial, innovative therapeutic products and by providing timely, evidence-based and authoritative information to allow consumers and health professionals to make informed decisions.
The TGA defines therapeutic product vigilance tools as tools designed to facilitate the collection and evaluation of information pertaining to the benefits and risks associated with the use of therapeutic products. The main product vigilance tools used by OPR are adverse event reports, Risk Management Plans and Periodic Safety Update Reports (PSURs).
Adverse event reports are reports of any untoward occurrence in a patient administered a medicine and which does not necessarily have a causal relationship with the medicine. An adverse event can therefore be any unfavourable and unintended sign, symptom, or disease temporally associated with the use of a medicine, whether or not considered related to this medicine.
RMPs provide a summary of the known important safety information about the therapeutic product, plans to identify and characterise known or potential safety concerns and plans to minimise any identified or potential safety risk. A full outline of the scope of RMPs is above (see Expert advisory committee). PSURs give an annual overview of the safety of the drug, including adverse events, a summary of the registration status of the drug world-wide, actions taken for safety reasons, the world-wide usage of the drug and an analysis of safety requirements. Sponsors must submit PSURs for at least three years after registration of a drug.
An important aspect of product vigilance is ensuring there are mechanisms to communicate safety information to both health professionals and consumers. To achieve this, the TGA publishes Australian Public Assessment Reports (AusPARs) about recently registered prescription medicines on the TGA website. The AusPAR outlines the findings of the TGA’s evaluation of a product including important effectiveness and safety information. The TGA also publishes the Product Information (PI) and Consumer Medicine Information (CMI) for prescription and some non-prescription medicines, Medicines Safety Update and alerts, which provide important information and recommendations about therapeutic goods.
THE HIGHLY SPECIALISED DRUGS PROGRAMME
Overview
The Australian Government provides funding for certain specialised medications under the Highly Specialised Drugs Programme. Highly specialised drugs (HSDs) are medicines for the treatment of chronic conditions which, because of their clinical use or other special features, are restricted to supply through public and private hospitals having access to appropriate specialist facilities. To prescribe these drugs as pharmaceutical benefit items, medical practitioners are required to be affiliated with these specialist hospital units. A general practitioner or non-specialist hospital doctor may prescribe HSDs to provide maintenance therapy under the guidance of the treating specialist.
The Australian Government provides funding for a HSD to be supplied to community based patients, not in-patients of public hospitals; i.e. persons who are day-admitted patients, outpatients and patients upon discharge.
The subsidies for drugs under this programme commences after approval has been given by the Australian Government. For HSDs prescribed through public and private hospitals, claiming and approval of authority prescriptions is administered by the Department of Human Services.
From 1 July 2013, public hospitals transitioned to online claiming arrangements for HSDs (with the exception of clozapine). This transition enables more efficient claiming processes and reduces the administrative burden on public hospitals. In addition, it enables data to be collected at the individual script level rather than summarised at the itemcode level, allowing easier comparison with private hospital data.
Table A: Highly Specialised Drugs—National Usage and Patient Report in Public Hospitals for the period January 2011 to December 2011
| Drug Name | Total Cost ($Aus) | Pack Numbers |
|---|---|---|
| ABACAVIR | $1,573,933 | 5,683 |
| ABACAVIR + LAMIVUDINE | $18,087,292 | 31,256 |
| ABACAVIR + LAMIVUDINE + ZIDOVUDINE | $1,455,722 | 1,658 |
| ABATACEPT | $8,423,165 | 16,154 |
| ADALIMUMAB | $1,351,408 | 793 |
| ADEFOVIR DIPIVOXIL | $4,124,100 | 7,421 |
| AMBRISENTAN | $7,626,514 | 1,864 |
| APOMORPHINE | $2,837,927 | 34,135 |
| ATAZANAVIR | $18,424,121 | 32,956 |
| AZACITIDINE | $9,217,270 | 16,729 |
| AZITHROMYCIN | $66,883 | 1,168 |
| BACLOFEN | $1,187,954 | 8,009 |
| BOSENTAN | $32,875,171 | 8,045 |
| CIDOFOVIR | $5,400 | 6 |
| CINACALCET | $6,611,120 | 14,465 |
| CLARITHROMYCIN | $112,908 | 2,098 |
| CLOZAPINE | $42,706,724 | 158,474 |
| CYCLOSPORIN | $9,693,191 | 120,431 |
| DARBEPOETIN ALFA | $52,611,173 | 88,973 |
| DARUNAVIR | $10,039,593 | 9,424 |
| DEFERASIROX | $13,519,820 | 16,276 |
| DEFERIPRONE | $624,724 | 1,411 |
| DESFERRIOXAMINE | $802,738 | 16,037 |
| DIDANOSINE | $350,454 | 1,271 |
| DORNASE ALFA | $10,126,752 | 8,578 |
| DOXORUBICIN HYDROCHLORIDE-PEGYLATED LIPOSOMAL | $148,895 | 239 |
| EFAVIRENZ | $5,032,726 | 10,902 |
| ELTROMBOPAG | $36,560 | 15 |
| EMTRICITABINE | $155,870 | 551 |
| ENFUVIRTIDE | $462,517 | 209 |
| ENTECAVIR | $11,193,542 | 26,661 |
| EPOETIN ALFA | $20,995,075 | 31,834 |
| EPOETIN BETA | $9,227,218 | 14,255 |
| EPOETIN LAMBDA | $107,736 | 171 |
| EPOPROSTENOL | $1,811,782 | 22,027 |
| ETANERCEPT | $2,572,834 | 2,608 |
| ETRAVIRINE | $3,520,591 | 5,634 |
| EVEROLIMUS | $6,397,008 | 9,743 |
| FILGRASTIM | $12,068,609 | 7,775 |
| FOSAMPRENAVIR | $550,466 | 1,462 |
| FOSCARNET | $1,188 | 3 |
| GANCICLOVIR | $112,014 | 380 |
| IBANDRONIC ACID | $453,667 | 1,329 |
| ILOPROST | $1,829,539 | 1,657 |
| INDINAVIR | $75,553 | 166 |
| INFLIXIMAB | $45,824,399 | 60,172 |
| INTERFERON ALFA-2A | $35,011 | 981 |
| INTERFERON ALFA-2B | $496,788 | 1,342 |
| INTERFERON GAMMA-1B | $471,089 | 346 |
| LAMIVUDINE | $4,040,646 | 21,074 |
| LAMIVUDINE + ZIDOVUDINE | $3,873,947 | 6,669 |
| LANREOTIDE | $6,161,662 | 3,384 |
| LANTHANUM | $1,732,821 | 4,763 |
| LENALIDOMIDE | $27,240,973 | 4,213 |
| LENOGRASTIM | $237,241 | 276 |
| LEVODOPA + CARBIDOPA ANHYDROUS | $127,792 | 89 |
| LOPINAVIR + RITONAVIR | $11,038,876 | 16,402 |
| MARAVIROC | $1,533,798 | 1,935 |
| METHOXY POLYETHYLENE GLYCOL-EPOETIN BETA | $4,677,379 | 7,571 |
| MYCOPHENOLATE | $19,958,297 | 96,292 |
| NATALIZUMAB | $26,936,210 | 13,213 |
| NEVIRAPINE | $8,811,745 | 31,874 |
| OCTREOTIDE | $12,294,556 | 10,696 |
| OMALIZUMAB | $249,345 | 579 |
| PAMIDRONATE DISODIUM | $1,240,631 | 4,658 |
| PEGFILGRASTIM | $49,237,207 | 25,578 |
| PEGINTERFERON ALFA-2A | $1,909,230 | 1,427 |
| PEGINTERFERON ALFA-2A (&) RIBAVIRIN | $29,278,215 | 17,889 |
| PEGINTERFERON ALFA-2B | $8,602 | 4 |
| PEGINTERFERON ALFA-2B (&) RIBAVIRIN | $9,877,876 | 5,238 |
| RALTEGRAVIR | $15,265,306 | 22,738 |
| RIFABUTIN | $42,020 | 286 |
| RITONAVIR | $1,841,951 | 47,521 |
| RITUXIMAB | $6,389,339 | 2,807 |
| ROMIPLOSTIM | $4,312,328 | 2,982 |
| SAQUINAVIR | $443,639 | 850 |
| SEVELAMER | $3,265,291 | 10,533 |
| SILDENAFIL | $1,489,525 | 1,590 |
| SIROLIMUS | $2,718,799 | 3,275 |
| SITAXENTAN | $2,744 | 1 |
| STAVUDINE | $188,052 | 453 |
| TACROLIMUS | $21,787,047 | 64,599 |
| TELBIVUDINE | $13,073 | 52 |
| TENOFOVIR | $11,052,302 | 22,737 |
| TENOFOVIR + EMTRICITABINE | $39,701,628 | 51,121 |
| TENOFOVIR + EMTRICITABINE + EFAVIRENZ | $44,233,573 | 35,714 |
| THALIDOMIDE | $7,803,113 | 15,828 |
| TIPRANAVIR | $174,005 | 162 |
| TOCILIZUMAB | $7,611,632 | 13,838 |
| VALACICLOVIR | $219,300 | 565 |
| VALGANCICLOVIR | $13,308,216 | 6,149 |
| ZIDOVUDINE | $313,044 | 1,444 |
| ZOLEDRONIC ACID | $13,609,580 | 30,239 |
| Grand Total | $794,289,288 | 1,413,083 |
SUPPLY OF PHARMACEUTICAL BENEFITS TO REMOTE AREA ABORIGINAL HEALTH SERVICES UNDER SECTION 100 OF THE NATIONAL HEALTH ACT
The S100 Supply Arrangements for Remote Area Aboriginal Health Services (AHSs) improve access to the PBS for clients of remote area AHSs under Section 100 of the National Health Act 1953.
Under these arrangements, clients of participating AHSs are able to receive PBS medicines directly from the AHS at the point of consultation, without the need for a normal prescription form, and without charge.
The eligibility criteria for participation in the program are given below.
Eligibility criteria
- The health service must have a primary function of meeting the health care needs of Aboriginal and Torres Strait Islander peoples.
- The clinic or other health care facility operated by the AHS from which pharmaceuticals are supplied to patients must be in a remote zone as defined in the Rural, Remote and Metropolitan Areas Classification 1991 Census Edition.
- The AHS must not be a party to an arrangement, such as a coordinated care trial, for which funds from the PBS have already been provided.
- The AHS must employ or be in a contractual relationship with health professionals
who are suitably qualified under relevant State/Territory legislation to supply all
medications covered by the Section 100 arrangements and undertake that all supply
of benefit items will be under the direction of such qualified persons.
- The clinic or other health care facility operated by the AHS from which pharmaceuticals are supplied must have storage facilities that will:
- prevent access by unauthorised persons;
- maintain the quality (eg chemical and biological stability and sterility) of the pharmaceutical; and
- comply with any special conditions specified by the manufacturer of the pharmaceutical.
Expenditure
In 2011, 173 remote area Aboriginal Health Services (AHS) were approved to participate in the S100 remote area AHS Program and 162 AHS actually received supply of PBS medicines in that period (note: although approved, some AHS may elect not to participate, or may participate on an intermittent basis). Participating AHS in 2011 were from remote locations in Northern Territory, Queensland, Western Australia, South Australia, New South Wales and Tasmania. S100 remote area AHS expenditure for PBS medicines in 2011 was $44.1 million (GST Inclusive).
HEALTH EXPENDITURE TRENDS
Table B (i): Total expenditure on pharmaceuticals and other medical non-durables as % total expenditure on health, TEH
| 2000 | 2001 | 2002 | 2003 | 2004 | 2005 | 2006 | 2007 | 2008 | 2009 | 2010 | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Australia | 14.8 | 15.1 | 14.5 | 15.0 | 14.8 | 14.3 | 14.3 | 14.3 | 14.6 | 14.7 | n.a. |
| Canada | 15.9 | 16.2 | 16.7 | 17.0 | 17.3 | 17.2 | 17.4 | 17.2 | 17.0 | 17.0 | 16.7 |
| France | 16.5 | 16.9 | 16.8 | 16.7 | 16.8 | 16.7 | 16.3 | 16.4 | 16.4 | 16.1 | 16.0 |
| Germany | 13.5 | 14.1 | 14.3 | 14.4 | 13.8 | 15.0 | 14.7 | 15.0 | 15.0 | 14.9 | 14.8 |
| New Zealand | n.a. | n.a. | n.a. | n.a. | 10.8 | 10.7 | 11.3 | 10.4 | 9.7 | 9.5 | 9.4 |
| United Kingdom | 14.2 | 13.9 | 13.5 | 13.5 | 13.3 | 12.8 | 12.3 | 12.1 | 11.8 | n.a. | n.a. |
| United States | 11.3 | 11.7 | 11.9 | 12.1 | 12.2 | 12.2 | 12.4 | 12.3 | 12.1 | 12.2 | 11.9 |
Table B (ii): Total expenditure on pharmaceuticals & other medical non-durables per capita, US$ purchasing power parity
| 2000 | 2001 | 2002 | 2003 | 2004 | 2005 | 2006 | 2007 | 2008 | 2009 | 2010 | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Australia | 336 | 361 | 370 | 400 | 425 | 426 | 453 | 480 | 503 | 541 | n.a. |
| Canada | 401 | 442 | 478 | 520 | 554 | 593 | 638 | 663 | 679 | 732 | 741 |
| France | 420 | 460 | 490 | 498 | 523 | 551 | 569 | 603 | 613 | 632 | 634 |
| Germany | 362 | 397 | 422 | 445 | 438 | 505 | 526 | 559 | 595 | 629 | 640 |
| New Zealand | n.a. | n.a. | n.a. | n.a. | 213 | 227 | 270 | 254 | 261 | 279 | 285 |
| United Kingdom | 260 | 278 | 296 | 313 | 337 | 346 | 363 | 366 | 369 | n.a. | n.a. |
| United States | 540 | 600 | 665 | 727 | 778 | 819 | 881 | 920 | 941 | 977 | 983 |
Table B (iii): Total expenditure on health as % gross domestic product
| 2000 | 2001 | 2002 | 2003 | 2004 | 2005 | 2006 | 2007 | 2008 | 2009 | 2010 | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Australia | 8.0 | 8.1 | 8.4 | 8.3 | 8.5 | 8.4 | 8.5 | 8.5 | 8.7 | 9.1 | n.a. |
| Canada | 8.8 | 9.3 | 9.6 | 9.8 | 9.8 | 9.8 | 10.0 | 10.0 | 10.3 | 11.4 | 11.4 |
| France | 10.1 | 10.2 | 10.6 | 10.9 | 11.0 | 11.2 | 11.0 | 11.0 | 11.0 | 11.7 | 11.6 |
| Germany | 10.4 | 10.5 | 10.7 | 10.9 | 10.7 | 10.8 | 10.6 | 10.5 | 10.7 | 11.7 | 11.6 |
| New Zealand | 7.6 | 7.7 | 8.0 | 7.9 | 8.0 | 8.4 | 8.8 | 8.5 | 9.3 | 10.0 | 10.1 |
| United Kingdom | 7.0 | 7.3 | 7.6 | 7.8 | 8.0 | 8.2 | 8.5 | 8.5 | 8.8 | 9.8 | 9.6 |
| United States | 13.7 | 14.3 | 15.1 | 15.7 | 15.8 | 15.8 | 15.9 | 16.2 | 16.6 | 17.7 | 17.6 |
Legend
n.a.: Not available (data not provided to the Organisation for Economic Co-operation
and Development (OECD) by these countries for these years)
Sources: OECD Health Data 2012—Version June 2012
(a) ‘Pharmaceuticals’ defined
The OECD definition of pharmaceuticals has been used, as described in its System of Health Accounts (SHA). The OECD defines pharmaceuticals as ‘pharmaceuticals and other medical non-durables dispensed to out-patients’, which comprises prescription medicines, over-the-counter medicines and other medical non-durables. Pharmaceuticals dispensed to, or used by admitted patients in hospital are not included.
Broadly speaking, these include medicinal preparations, branded and generic medicines, drugs, patent medicines, serums and vaccines, vitamins and minerals, and oral contraceptives and a wide range of medical non-durable goods, which are either single use items, for example bandaids and condoms, or have limited re-usage, for example, bandages.
Prescribed medicines are medicines exclusively sold to customers with a medical voucher, irrespective of whether it is covered by public or private funding and include branded and generic products. In the SHA, this includes the full price with a breakdown for cost-sharing.
Expenditure by private households or over-the-counter medicines (OTC medicines) is included in pharmaceutical expenditure.
Other medical non-durables comprise items such as bandages, elastic stockings, incontinence articles, condoms and other mechanical contraceptive devices.
Pharmaceutical expenditure is also reported in Health Expenditure Australia 2009-10. In this report, the Australian Institute of Health and Welfare (AIHW) splits medication expenditure into two categories, ‘benefit paid pharmaceuticals’ and ‘all other medications’.
‘Benefit paid pharmaceuticals’ are pharmaceuticals in the Pharmaceutical Benefits Scheme (PBS) and the Repatriation Pharmaceutical Benefits Scheme (RPBS) for which the Australian Government paid a benefit.
‘All other medications’ are medications for which no PBS or RPBS benefit was paid and includes the following components, which are reported separately:
- pharmaceuticals listed in the PBS or RPBS, where the total costs are equal to, or less than, the statutory patient contribution for the class of patient concerned (‘under co-payment’ drugs);
- medicines dispensed through private prescriptions for items not listed in the PBS or RPBS or which do not meet PBS criteria for dispensing through the PBS; and
- over-the-counter medicines such as aspirin, cough and cold medicines, vitamins and minerals, some herbal and other complementary medicines and medical non-durable goods, as listed above.
Under the AIHW definitions used in Health expenditure Australia, highly specialised drugs are included as part of hospital expenditure, not as part of medications expenditure. Under the OECD definition, highly specialised drugs are included in ‘Pharmaceutical’ expenditure.
(b) Health expenditure recorded here is according to the OECD definition and excludes health research expenditure.
DRUG UTILISATION TRENDS
Anatomical Therapeutic Chemical (ATC) classification index with Defined Daily Doses (DDDs) 2012 is used in all statistics published in this edition (refer to WHO collaborating Centre for Drug Statistics Methodology, ATC classification index with DDDs 2012).
Listed below are the prescription counts for 2009, 2010 and 2011 by ATC anatomical main group. The data from the two sources are enumerated separately. Table C (i) shows subsidised prescriptions (PBS/RPBS) and Table C (ii) shows the estimate of nonsubsidised prescriptions (Survey).
Tables C: Prescription numbers by ATC groups
Table C (i): Subsidised prescriptions (PBS/RPBS)
| ATC Group | 2009 | 2010 | 2011 |
|---|---|---|---|
| (A) Alimentary Tract | 28,400,464 | 28,934,869 | 28,741,866 |
| (B) Blood and blood forming | 8,640,003 | 8,961,287 | 9,011,672 |
| (C) Cardiovascular system | 67,302,312 | 69,501,887 | 71,052,963 |
| (D) Dermatologicals | 2,980,367 | 3,044,864 | 3,100,758 |
| (G) Genitourinary system | 3,172,962 | 3,016,920 | 3,054,098 |
| (H) Hormonal preparations | 2,906,146 | 3,028,197 | 3,107,836 |
| (J) Antiinfectives | 13,546,581 | 13,603,470 | 14,086,713 |
| (L) Antineoplastic | 2,098,520 | 2,282,769 | 2,400,647 |
| (M) Musculo-skeletal | 9,262,036 | 9,044,116 | 8,871,353 |
| (N) Nervous system | 38,838,056 | 41,109,237 | 43,141,389 |
| (P) Antiparasitic products | 583,591 | 619,618 | 662,647 |
| (R) Respiratory system | 10,544,777 | 10,924,181 | 11,222,832 |
| (S) Sensory Organs | 8,718,735 | 8,826,316 | 8,839,667 |
| (V) Various | 646,443 | 627,657 | 614,535 |
| Other | 209,959 | 234,134 | 237,869 |
| Total | 197,850,952 | 203,759,522 | 208,146,845 |
Table C (ii): Estimated non-subsidised prescriptions (Survey)
| ATC Group | 2009 | 2010 | 2011 |
|---|---|---|---|
| (A) Alimentary Tract | 5,394,413 | 5,443,423 | 6,778,526 |
| (B) Blood and blood forming | 826,678 | 799,144 | 795,439 |
| (C) Cardiovascular system | 13,069,382 | 12,749,658 | 12,459,359 |
| (D) Dermatologicals | 2,998,874 | 3,090,690 | 3,107,951 |
| (G) Genitourinary system | 7,319,091 | 7,241,951 | 7,237,305 |
| (H) Hormonal preparations | 1,600,906 | 1,673,213 | 1,709,388 |
| (J) Antiinfectives | 15,530,330 | 15,262,828 | 15,802,547 |
| (L) Antineoplastic | 118,612 | 134,160 | 150,004 |
| (M) Musculo-skeletal | 3,168,661 | 3,088,987 | 3,106,030 |
| (N) Nervous system | 12,106,849 | 12,179,569 | 12,804,813 |
| (P) Antiparasitic products | 691,724 | 671,856 | 688,164 |
| (R) Respiratory system | 2,696,789 | 2,648,049 | 2,586,732 |
| (S) Sensory Organs | 2,400,492 | 2,294,943 | 2,155,669 |
| (V) Various | 19,842 | 21,220 | 26,030 |
| Other | 220,046 | 232,186 | 245,644 |
| Total | 68,162,689 | 67,531,877 | 69,653,601 |
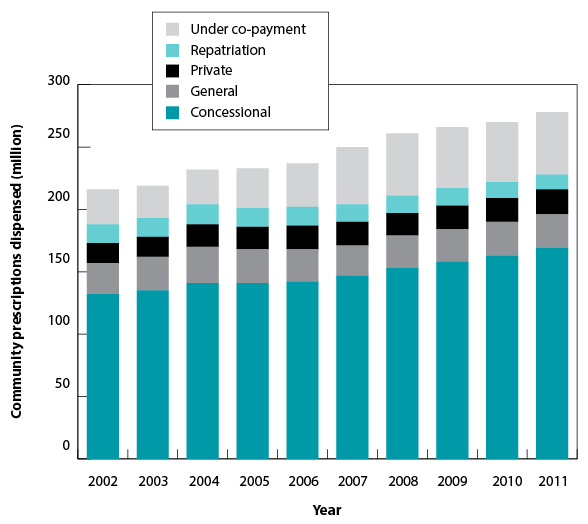
Estimated changes from 2002 to 2011 in the number of prescriptions dispensed under the PBS (concession and general), RPBS, under co-payment and private categories, are presented in Figure C.
Figure C: Number of prescriptions by type of service
Most commonly used drugs in the Australian community for 2011
Table D shows the top 10 drugs dispensed in the Australian community by DDDs/1000 population/day, which adjusts for the quantity dispensed per prescription. This DDDs/1000/day information shows both the subsidised (PBS/RPBS) and non-subsidised (Guild survey) components, as well as total community use. Changes and alterations from the previous years are also shown.
Table D: Top 10 drugs by defined daily dose/thousand population/day, 2011 (including the contribution of constituents of combination products)
| Drug | PBS/RPBS | Guild Survey | Total |
|---|---|---|---|
| ATORVASTATIN | 85.043 | 0.139 | 85.182 |
| IRBESARTAN | 33.007 | 10.187 | 43.194 |
| PERINDOPRIL | 31.124 | 11.129 | 42.253 |
| ROSUVASTATIN | 36.158 | 0.067 | 36.225 |
| RAMIPRIL | 25.910 | 8.813 | 34.723 |
| CANDESARTAN | 26.960 | 5.265 | 32.225 |
| PARACETAMOL | 28.990 | 2.559 | 31.549 |
| AMLODIPINE | 22.105 | 3.991 | 26.096 |
| SIMVASTATIN | 23.654 | 0.672 | 24.325 |
| ESOMEPRAZOLE | 21.454 | 2.700 | 24.154 |
Changes from 2010:
| UP: | Rosuvastatin (5 |
| Amlodipine (9 |
|
| DOWN: | Ramipril (4 |
| Simvastatin (8 |
The top 10 drugs dispensed in the Australian community in 2011, ranked by prescription count, are shown in table E. Table F ranks the 2011 top 10 drugs by total cost to Australia, i.e. subsidised prescriptions only (total cost is the sum of patient contribution and cost to Government).
Table E: Top 10 drugs by prescription counts, 2011
| Drug | PBS/RPBS | Guild Survey | Total Community use |
|---|---|---|---|
| ATORVASTATIN | 11,100,222 | 20,422 | 11,120,644 |
| ESOMEPRAZOLE | 5,890,893 | 1,088,070 | 6,978,963 |
| ROSUVASTATIN | 6,595,316 | 12,321 | 6,607,637 |
| AMOXYCILLIN | 2,592,120 | 3,670,480 | 6,262,600 |
| PARACETAMOL | 5,181,539 | 276,598 | 5,458,137 |
| PERINDOPRIL | 3,971,080 | 1,468,112 | 5,439,192 |
| CEFALEXIN | 2,740,226 | 2,380,837 | 5,121,063 |
| PANTOPRAZOLE | 3,700,004 | 844,942 | 4,544,946 |
| METFORMIN HYDROCHLORIDE | 3,432,146 | 1,092,602 | 4,524,748 |
| AMOXYCILLIN with CLAVULANIC ACID | 2,014,812 | 2,376,545 | 4,391,357 |
Changes from 2010:
| UP: | Rosuvastatin (5 |
| Paracetamol (6 |
|
| Pantoprazole (10 |
|
| DOWN: | Amoxycillin (3 |
| Perindopril (4 |
|
| IN: | Amoxycillin with Clavulanic acid (13 |
| OUT: | Simvastatin (8 |
Table F: Top 10 PBS/RPBS drugs by total cost to Australia, 2011
| Drug | PBS/RPBS DDD/1000/DAY |
PBS/RPBS Scripts |
Total Cost ($) |
|---|---|---|---|
| ATORVASTATIN | 85.043 | 11,100,222 | 784,927,075 |
| ROSUVASTATIN | 36.158 | 6,595,316 | 467,425,285 |
| RANIBIZUMAB | 160,813 | 342,880,160 | |
| ESOMEPRAZOLE | 21.454 | 5,890,893 | 230,060,391 |
| SALMETEROL and FLUTICASONE | 3,124,942 | 221,699,121 | |
| ADALIMUMAB | 0.368 | 107,020 | 192,726,034 |
| CLOPIDOGREL | 11.179 | 2,706,737 | 187,035,815 |
| OLANZAPINE | 3.084 | 967,080 | 174,280,242 |
| SIMVASTATIN | 23.654 | 4,087,534 | 161,894,597 |
| VENLAFAXINE | 12.150 | 2,598,159 | 140,328,907 |
No information on cost for private and under co-payment prescriptions is available.
Changes from 2010:
| UP: | Salmeterol and Fluticasone (6 |
| Adalimumab (9 |
|
| DOWN: | Clopidogrel (5 |
| Simvastatin (7 |
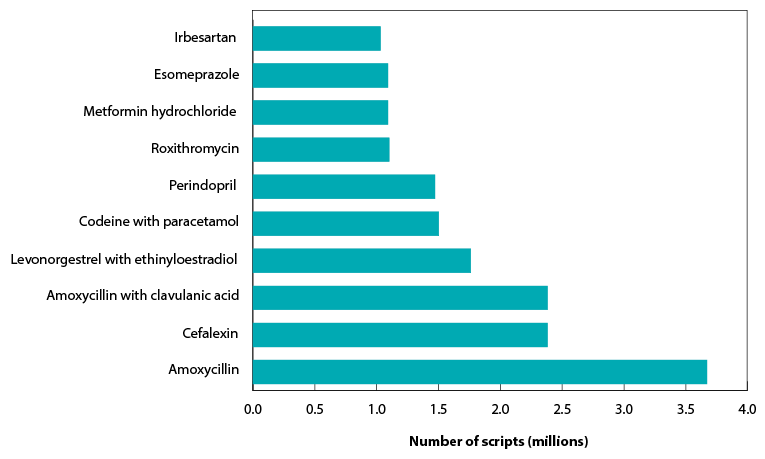
Figure D shows the top 10 subsidised drugs dispensed in 2011.
Figure D: Top 10 subsidised drugs dispensed in 2011
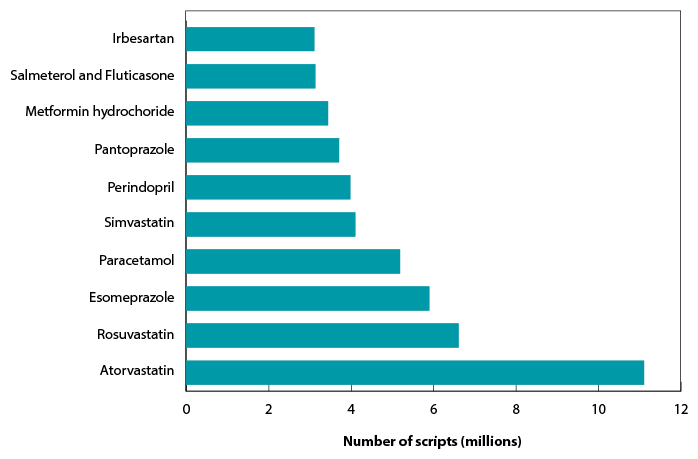
Figure E represents the top 10 non-subsidised drugs for 2011.
Figure E: Top 10 non-subsidised drugs dispensed in 2011
Nurse Prescribing
From 1 November 2010, authorised nurse practitioners can write PBS/RPBS prescriptions listed in the PBS Schedule with a flag of “NP”. Medicines without this flag are not to be prescribed by authorised nurse practitioners. Refer to Schedule of Pharmaceutical Benefits available on the PBS website for details of subsidised medicines.
Information for how nurse practitioners can become authorised PBS prescribers is available from the Department of Human and Services website.
Table G shows the top 10 PBS/RPBS drugs prescribed by nurse practitioners and dispensed in 2011. Table H ranks the 2011 top 10 drugs prescribed by total cost.
Table G: Nurse Practitioner—Top 10 PBS/RPBS drugs by prescription counts, 2011
| Drug | PBS/RPBS Scripts |
|---|---|
| CEPHALEXIN | 1,137 |
| ATORVASTATIN | 1,013 |
| AMOXYCILLIN with CLAVULANIC ACID | 750 |
| CODEINE PHOSPHATE with PARACETAMOL | 732 |
| OXYCODONE HYDROCHLORIDE | 713 |
| PARACETAMOL | 685 |
| AMOXYCILLIN | 635 |
| FLUCLOXACILLIN | 549 |
| ROSUVASTATIN | 536 |
| FLUTICASONE PROPIONATE with SALMETEROL XINAFOATE | 523 |
Table H: Nurse Practitioner—Top 10 PBS/RPBS drugs prescribed by total cost, 2011
| Drug | Total Cost ($) |
|---|---|
| ATORVASTATIN | 72,512 |
| RISPERIDONE | 69,855 |
| PANCREATIC EXTRACT | 67,913 |
| OLANZAPINE | 46,693 |
| QUETIAPINE | 37,390 |
| ROSUVASTATIN | 37,185 |
| FLUTICASONE PROPIONATE with SALMETEROL XINAFOATE | 35,858 |
| VALACICLOVIR | 26,348 |
| INSULIN GLARGINE | 24,006 |
| POSACONAZOLE | 23,109 |
Optometrist Prescribing
Since 1 January 2008, authorised optometrists are able to prescribe medicines listed in the optometrist section of the Pharmaceutical Benefits Schedule. Refer to Pharmaceutical Benefits Schedule for a list of medicines that can be prescribed by authorised optometrists.
Table I shows the top 10 PBS/RPBS drugs prescribed by optometrists and dispensed in 2011. Table H ranks the 2011 top 10 drugs prescribed by total cost.
Table I: Optometrist—Top 10 PBS/RPBS drugs by prescription counts, 2011
| Drug | PBS/RPBS Scripts |
|---|---|
| LATANOPROST | 8,022 |
| POLYETHYLENE GLYCOL 400 with PROPYLENE GLYCOL | 7,754 |
| CHLORAMPHENICOL | 5,781 |
| FLUOROMETHOLONE | 4,864 |
| CARMELLOSE SODIUM | 3,635 |
| HYPROMELLOSE | 2,244 |
| LATANOPROST with TIMOLOL MALEATE | 2,031 |
| HYPROMELLOSE with CARBOMER 980 | 1,983 |
| PARAFFIN | 1,760 |
| FLUOROMETHOLONE ACETATE | 1,704 |
Table J: Optometrist—Top 10 PBS/RPBS drugs prescribed by total cost, 2011
| Drug | Total Cost ($) |
|---|---|
| LATANOPROST | 338,041 |
| LATANOPROST with TIMOLOL MALEATE | 94,649 |
| POLYETHYLENE GLYCOL 400 with PROPYLENE GLYCOL | 91,663 |
| CHLORAMPHENICOL | 60,839 |
| CARMELLOSE SODIUM | 60,402 |
| BIMATOPROST | 57,479 |
| FLUOROMETHOLONE | 51,607 |
| TRAVOPROST | 41,466 |
| PARAFFIN | 35,296 |
| BIMATOPROST with TIMOLOL MALEATE | 27,074 |
TABLES IN THE AUSTRALIAN STATISTICS ON MEDICINES
The data are presented in two major tables. Table 1 provides an estimate of the 2011 community (i.e. subsidised and non-subsidised) prescription numbers, together with the costs for PBS-listed drugs, which include an estimate of the cost of under co-payment PBS prescriptions. Cost information on the dispensing of private prescriptions is not available. The defined daily dose (DDD), where available, is also included for the drugs covered in the report.
Table 2 includes community prescription drug use, in DDDs/1000 population/day, for the years 2009, 2010 and 2011. In this edition, DDDs/1000 population/day for combination products is also reported in terms of DDDs of each constituent. One main advantage of reporting combinations as if they were administered as two or more single component products is that total DDDs remain constant as patients switch to combination products, if the amounts of constituent drugs consumed by patients remain the same.
Note that not all combination products are included in table 2. Combination drugs will only be reported in terms of DDD of each constituent where:
- the constituent drugs were available as plain drugs on the PBS;
- the combination was a frequently prescribed form;
- the constituent drugs had independent actions; and
- the WHO Defined Daily Dose was consistent across the various formulations of the constituent drugs.
Anatomical Therapeutic Chemical (ATC) classification index with Defined Daily Doses (DDDs) 2012 is used in statistics published in this edition (refer to WHO collaborating Centre for Drug Statistics Methodology, ATC classification index with DDDs 2012).
References
(1) Edmonds DJ, Dumbrell DM, Primrose JG, McManus P, Birkett DJ, Demirian V. Development of an Australian drug utilisation database: a report from the Drug Utilisation Subcommittee of the Pharmaceutical Benefits Advisory Committee, PharmacoEconomics 1993; 3(6): 427432.
(2) Hurley SF, McNeil JJ. Drugcoding systems: why so many?Med J Aust 1989; 151: 308.
(3) Nordic Council on Medicines. Nordic Statistics on Medicines 1987-1989. NLN publication number 3, Uppsala, Sweden, 1990.
(4) McManus P. Drug utilisation (letter)Med J Aust 1993; 158: 724.
CAVEATS
It needs to be borne in mind that these utilisation data do not include a large proportion of public hospital drug usage, over the counter purchases (except for S3 Recordable), or the supply of highly specialised drugs to outpatients through public hospitals, under Section 100 of the National Health Act 1953. Some extemporaneously prepared items may also not be included.
Comments on classifications, omissions or errata appearing in this edition of the Australian Statistics on Medicines may be sent to:
Alicia Segrave
Secretary
Drug Utilisation Sub-Committee (DUSC)
Department of Health
GPO Box 9848
CANBERRA ACT 2601.
e-mail: DUSC@health.gov.au
GLOSSARY OF TERMS
| Actu | Actuated |
| Adhes | Adhesive |
| Admin | Administration |
| Aero | Aerosol |
| Amp(s) | Ampoule(s) |
| Applic | Applicator |
| Aqu | Aqueous |
| Breth | Breath |
| Calc | Calcium |
| Cap(s) | Capsule(s) |
| Cart | Cartridge |
| CD | Controlled delivery |
| Chew | Chewable |
| Clean | Cleansing |
| Coat | Coated |
| Co | Compound |
| Conc | Concentrated |
| Cont | Contained |
| CR | Controlled release |
| Crm | Cream |
| Crush | Crushable |
| D | Dose |
| Dev | Device |
| Diag | Diagnostic |
| Dil | Diluted |
| Disp | Dispersable |
| Dres | Dressing |
| Drp | Drops |
| Ds | Doses |
| Dust | Dusting |
| Efferv | Effervescent |
| Elx | Elixir |
| Enter | Enteric |
| Emulsif | Emulsifying |
| Equiv | Equivalent |
| Extend | Extended |
| Ferr | Ferrous |
| Gran | Granules |
| Inf | Infusion |
| Inhal | Inhalation |
| Inj(s) | Injection(s) |
| Inrt | Inert |
| Ins | Insert |
| Intracav | Intracavernosal |
| Intranas | Intranasal |
| Insuff | Insufflator |
| Irrig | Irrigation |
| Jel | Jelly |
| Linct | Linctus |
| Lin | Liniment |
| Liq | Liquid |
| Loz | Lozenge |
| Ltn | Lotion |
| Metronid | Metronidazole |
| Mixt | Mixture |
| Nas | Nasal |
| Nebu | Nebuliser |
| Not< | Not less than |
| Oint | Ointment |
| Ophth | Ophthalmic |
| Paed | Paediatric |
| Pdr | Powder |
| Pell(s) | Pellet(s) |
| Pess | Pessary |
| Phos | Phosphorus |
| Pot | Potassium |
| Prep | Preparation |
| Press | Pressurised |
| Prot | Protective |
| Pst | Paste |
| Reag | Reagent |
| Rel | Release |
| Requ | Required |
| Sach(s) | Sachet(s) |
| SF | Sugar free |
| Sng | Single |
| Sod | Sodium |
| Sol | Soluble |
| Soln | Solution |
| Solv | Solvent |
| Spr | Spray |
| Ster | Sterile |
| Sulph | Sulphate |
| Suppl | Supplement |
| Suppos | Suppository |
| Supres | Suppression |
| Susp | Suspension |
| Sust | Sustained |
| Syrp | Syrup |
| Syrng | Syringe |
| Tab(s) | Tablet(s) |
| Td | Transdermal |
| Tinct | Tincture |
| Top | Topical |
| Unt(s) | Unit(s) |
| wps | Wipes |
Weights and Measures
| cm | centimetre(s) |
| E | unit(s) |
| g | gram(s) |
| kg | kilogram(s) |
| iu | international unit |
| L | litre(s) |
| m | metre(s) |
| ME | million units |
| mm | millimetre(s) |
| mg | milligram(s) |
| mL | millilitre(s) |
| mmol | millimole |
| TE | thousand units |
| ug | micrograms(s) |
ATC & DDD ADDITIONS AND ALTERATIONS
Alterations in ATC classification
| Drug/drug group | Previous ATC code | New ATC code |
|---|---|---|
| C1-inhibitor, plasma derived* | B02AB03 | B06AC01 |
| Icatibant | C01EB19 | B06AC02 |
| Alitretinoin | D11AX19 | D11AH04 |
*New 5th level name
Alterations in ATC level classification
| ATC code | Previous ATC level name | New ATC level name |
|---|---|---|
| A12AX | Calcium, combinations with other drugs | Calcium, combinations with vitamin D and/or other drugs |
| B06AC | Drugs used in hereditary angioedema | |
| D11AH | Agents for atopic dermatitis, excluding corticosteroids | Agents for dermatitis, excluding corticosteroids |
| G04CX01 | Pygeum africanum | Prunus africanae cortex |
| G04CX02 | Serenoa repens | Sabalis serrulatae fructus |
| H01CB | Antigrowth hormones | Somatostatin and analogues |
| J07AH03 | Meningococcus, bivalent purified polysaccharides antigen | Meningococcus A,C, bivalent purified polysaccharides antigen |
| J07AH04 | Meningococcus, tetravalent purified polysaccharides antigen | Meningococcus A, C, Y, W - 135, tetravalent purified polysaccharides antigen |
| J05AH08 | Meningococcus, tetravalent purified polysaccharides antigen conjugated | Meningococcus A, C, Y, W - 135, tetravalent purified polysaccharides antigen conjugated |
| L02BG | Enzyme inhibitors | Aromatase inhibitors |
| N05CM09 | Valerian | Valerianae radix |
| N06DX02 | Ginkgo biloba | Ginkgo folium |
| R05X | Other cold combination preparations | Other cold preparations |
Allocation of new ATC codes
| New ATC Code | ATC level name |
|---|---|
| A02BC06 | Dexlansoprazole |
| A10BD10 | Metformin and saxagliptin |
| A10BD11 | Metformin and linagliptin |
| A10BX09 | Dapagliflozin |
| A16AX08 | Teduglutide |
| B01AA12 | Fluindione |
| B02BD10 | Von willebrand factor |
| B02BD11 | Catridecacog |
| B06AC03 | Ecallantide |
| B06AC04 | Conestat alfa |
| C05CX03 | Hippocastani semen |
| C09BB07 | Ramipril and amlodipine |
| C09CA09 | Azilsartan medoxomil |
| C09DB06 | Losartan and amlodipine |
| C10AX11 | Mipomersen |
| D03AX11 | Tetrachlorodecaoxide |
| D06BB12 | Sinecatechins |
| G03DB08 | Dienogest |
| G04BX15 | Pentosan polysulfate sodium |
| H01AC06 | Tesamorelin |
| H01CB05 | Pasireotide |
| J01CA19 | Aspoxicillin |
| J01DC12 | Cefminox |
| J01DC13 | Cefbuperazone |
| J01DC14 | Flomoxef |
| J01DF02 | Carumonam |
| J01GB13 | Bekanamycin |
| J05AE11 | Telaprevir |
| J05AG05 | Rilpivirine |
| J06BB17 | Motavizumab |
| J07AH09 | Meningococcus B, multicomponent vaccine |
| L01CD04 | Cabazitaxel |
| L01XC11 | Ipilimumab |
| L01XC12 | Brentuximab vedotin |
| L01XE14 | Bosutinib |
| L01XE15 | Vemurafenib |
| L01XE16 | Crizotinib |
| L01XE17 | Axitinib |
| L01XE18 | Ruxolitinib |
| L01XX42 | Panobinostat |
| L02BX03 | Abiraterone |
| L03AX17 | Sipuleucel-T |
| L04AC10 | Seculinumab |
| L04AX05 | Pirfenidone |
| M01AC56 | Meloxicam, combinations |
| M01AE56 | Naproxen and misoprostol |
| N03AX22 | Perampanel |
| N05AX14 | Iloperidone |
| N06AX24 | Vilazodone |
| N06AX25 | Hyperici herba |
| N06DA52 | Donepezil and memantine |
| N07BC05 | Levomethadone |
| N07XX08 | Tafamidis |
| P03AX05 | Dimeticone |
| R06AA59 | Doxylamine, combinations |
| S01LA05 | Aflibercept |
| S02DA04 | Cinchocaine |
Allocation of new DDDs
| ATC level name | ATC code | New DDD |
|---|---|---|
| A03AE04 | Prucalopride | 2 mg O |
| A16AB10 | Velaglucerasa alfa | 300 U P |
| B01AC24 | Ticagrelor | 0.18 g O |
| B02BX05 | Eltrombopag | 50 mg O |
| B06AC04 | Conestat alfa | 3.5 TU P |
| C01BG11 | Vernakalant | 0.2 g P |
| G03DB08 | Dienogest | 2 mg O |
| G03GA09 | Corifollitropin alfa | 0.15 mg P |
| H01CA03 | Histrelin | 0.137 g2) implant |
| J01CA19 | Aspoxicillina | 4 g P |
| J01DC12 | Cefminox | 4 g P |
| J01DC13 | Cefbuperazone | 2 g P |
| J01DC14 | Flomoxef | 2 g P |
| J01DF01 | Aztreonam | 0.225 g Inhal. solution |
| J01DF02 | Carumonam | 2 g P |
| J01GB10 | Ribostamycin | 1 g P |
| J01GB11 | Isepamicin | 0.4 g P |
| J01GB13 | Bekanamycin | 0.6 g P |
| L03AX15 | Mifamurtide | 0.7 mg P |
| L04AA27 | Fingolimod | 0.5 mg O |
| M01AB51 | Indomethacin, combinations | 0.1 g1) O,R |
| N02AX06 | Tapentadol | 0.4 g O |
| N02BG07 | Flupirtine | 0.4 g O |
| N05AH05 | Asenapine | 20 mg O |
| N06AX23 | Desvenlafaxine | 50 mg O |
| N07XX07 | Fampridine | 20 mg O |
| R03AC18 | Indacaterol | 0.15 mg Inhal. powder |
| R03DX07 | Roflumilast | 0.5 mg O |
1) refers to indomethacin
2) DDD assigned according to the total content of the implant
Full details on current ATC coding and defined daily doses (DDDs) can be obtained from the DUSC Secretary, Department of Health, GPO Box 9848, Canberra ACT 2601, or direct from the coordinating body: the WHO Collaborating Centre for Drug Statistics Methodology, Norwegian Institute of Public Health, PO BOX 4404 Nydalen 0403 Oslo Norway, or at the WHO website.




